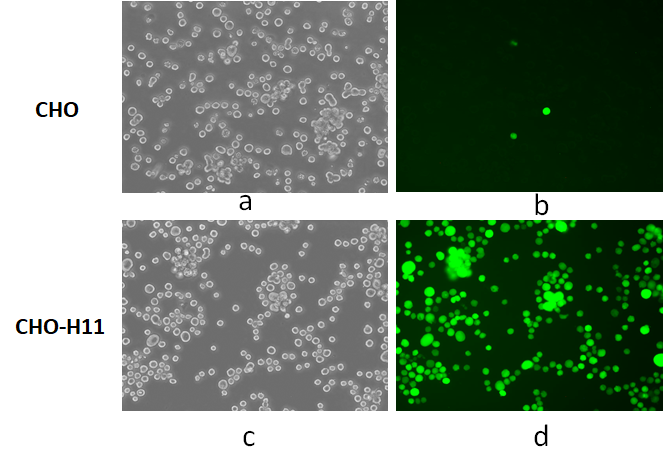
The traditional CHO antibody production method is very inefficient because it involves random gene insertion and forced amplification of transgenes. Many factors adversely affect bioproduction levels. These factors include insertion sites effects (position effects), DNA repeat-induced gene silencing and genomic instability, altered regulation or interruption of endogenous genes.
Thus, the goals of the CHO antibody production field are: a defined genomic locus with guaranteed high levels of expression, high integration efficiency and easy screening that is reproducible for any candidate protein. In addition, it is crucial that the cell line is viable and stably reproduced.
Applied StemCell has met and exceeded these goals with its proprietary TARGATT™ technology in CHO cell lines. Applied StemCell (ASC) is proud to announce the launch of our CHO Bioproduction cell line technology. ASC has created Master TARGATT™ CHO cell lines for the rapid creation of new lines for high level protein and antibody expression. Applied StemCell’s TARGATT™ CHO master cell lines yield >2.5g/L of recombinant proteins in a 2-week fed-batch shake flask expression system and is easily scalable for large scale bioproduction.