Newsletter
Efficacy Service with HU Models
Based on our world-class animal model genetic engineering technologies, Applied StemCell offers a comprehensive suite of downstream assays in mouse and rat models to validate your animal models and for drug screening. We have a multidisciplinary team of expert scientists who can design a comprehensive project plan to fit any requirement/stage of your research pipeline and for drug discovery and screening.
- Designing and engineering research animal disease models
- Adoptive transfers/transplantation
- In vivo functional screening assays
- End-of-study in vitro assays
- Antibody efficacy testing studies for immunotherapy for cancer and autoimmune disorders
Products and Services
Technical Details
Our project portfolio also comes with dedicated project managers for scheduling, timely communication and completion of your projects.
Below is a selected list of assays available for your in vivo assessments:
1. Disease Model Generation:
- Genetically modified mouse and rat models
- Adoptive cell transfer, teratomas
- Surgically/ drug induced models
2. In Vivo Assays:
- Behavioral assessments: cognition & locomotor activity
- Automated in vivo measurements: ECG, EEG, EMG
- In vivo pharmacokinetics
3. In Vitro / Postmortem Assays:
- Electrophysiology: neuronal & cardiac assays; patch-clamp, MEA
- Tissue collection and end-of-study analyses: western blots, immunohistochemistry, RT-PCR
Don’t see an assay / measurement? Please contact us to discuss your requirements with one of our technical experts.
Applications: Functional genomics, disease modeling, target identification and validation for drug discovery and screening, and many more.
Available Humanized Mouse Models
I. Human Immune System Reconstruction Models
II. Immune Checkpoint Humanized Models
I. Human Immune System Reconstruction Models
- Mouse Model Development: Transplantation of human-derived cell into mouse
II. Immune Checkpoint Humanized Models
- Mouse Model Development: human proteins expressed in mouse
- Humanized PD-1 Mouse
- Huamnized hCTLA4 Mouse
- Humanized OX40 Mouse
- Humanized LAG3 Mouse
Case Studies
1. Antibody Efficacy Testing in Humanized Immune Checkpoint Mouse Models
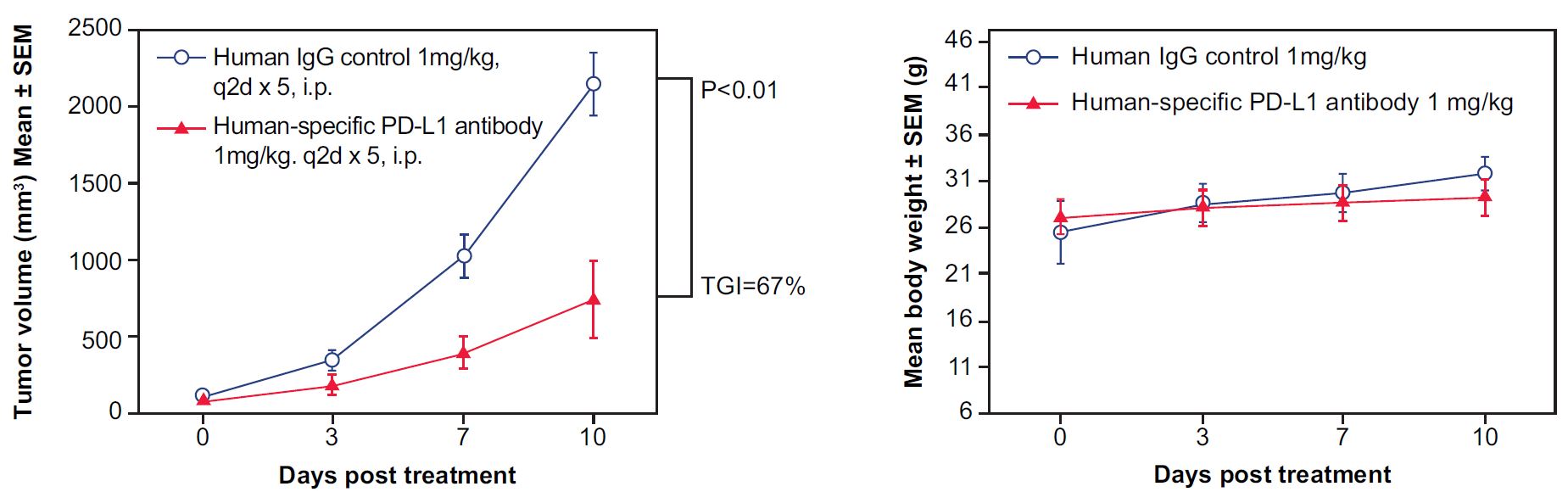
Figure 1. In vivo validation of anti-tumor efficacy in a MC38 tumor-bearing model of humanized PD-L1 mice. Mouse colon cancer cells MC38 engineered to express human PD-L1 were implanted subcutaneously into homozygous, humanized PD-L1 mice. The mice were randomly assigned into two groups when the tumor volume reached 100 mm3, with one group receiving human IgG as a control and the other receiving a human-specific, PD-L1 antibody (n=5). The human PD-L1 blocking antibody significantly inhibited tumor growth in the homozygous humanized mice (Left) without affecting overall body weight (Right), suggesting that the humanized PD-L1 mice represent an ideal model for evaluating the efficacy of therapeutic antibodies targeting human PD-L1. (TGI: tumor growth inhibition; 67%; p < 0.001), demonstrating that the humanized PD-L1 mice are a good in vivo model for validating the efficacy of antibodies targeting human PD-L1.
2. Cardiac Ion Channel Safety Screening Using Manual Patch Recording
Screening for potential cardiotoxicity of novel drug candidates that modulate key ion channels. Utilizing our expertise in patch-clamp electrophysiology, drugs can be screened against an array of ion channels including recombinant human ether-a-go-go (hERG), Nav1.5, Cav1.2, and human iPSC-derived cardiomyocytes.
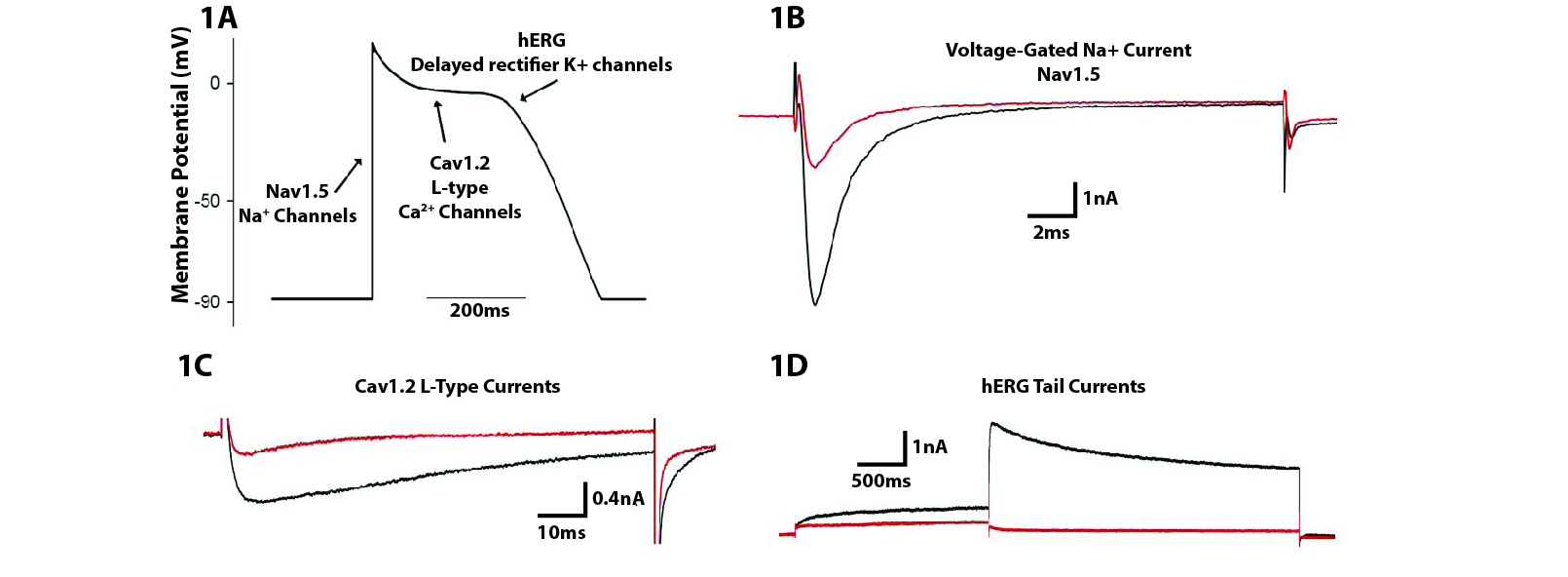
Figure 2. (A) Several ion channels involved in the cardiac action potential are implicated in QT interval prolongation. (B-D) Example recordings showing drug inhibition of human Nav1.5, Cav1.2 and hERG currents. Black traces represent control currents and red traces show currents in the presence of representative inhibitory drugs.
3. Functional Screening Assays to Test Neurological Endpoints Using Patch Clamp Recording
Electrophysiological Recordings in Rodent Brain Slices for disease model validation and drug efficacy/safety. Standard electrophysiological recordings of acutely prepared brain slices are used to measure changes in membrane potential, cell excitability, spike firing and synaptic neurotransmission, including long-term potentiation/ depression (LTP/LTD).
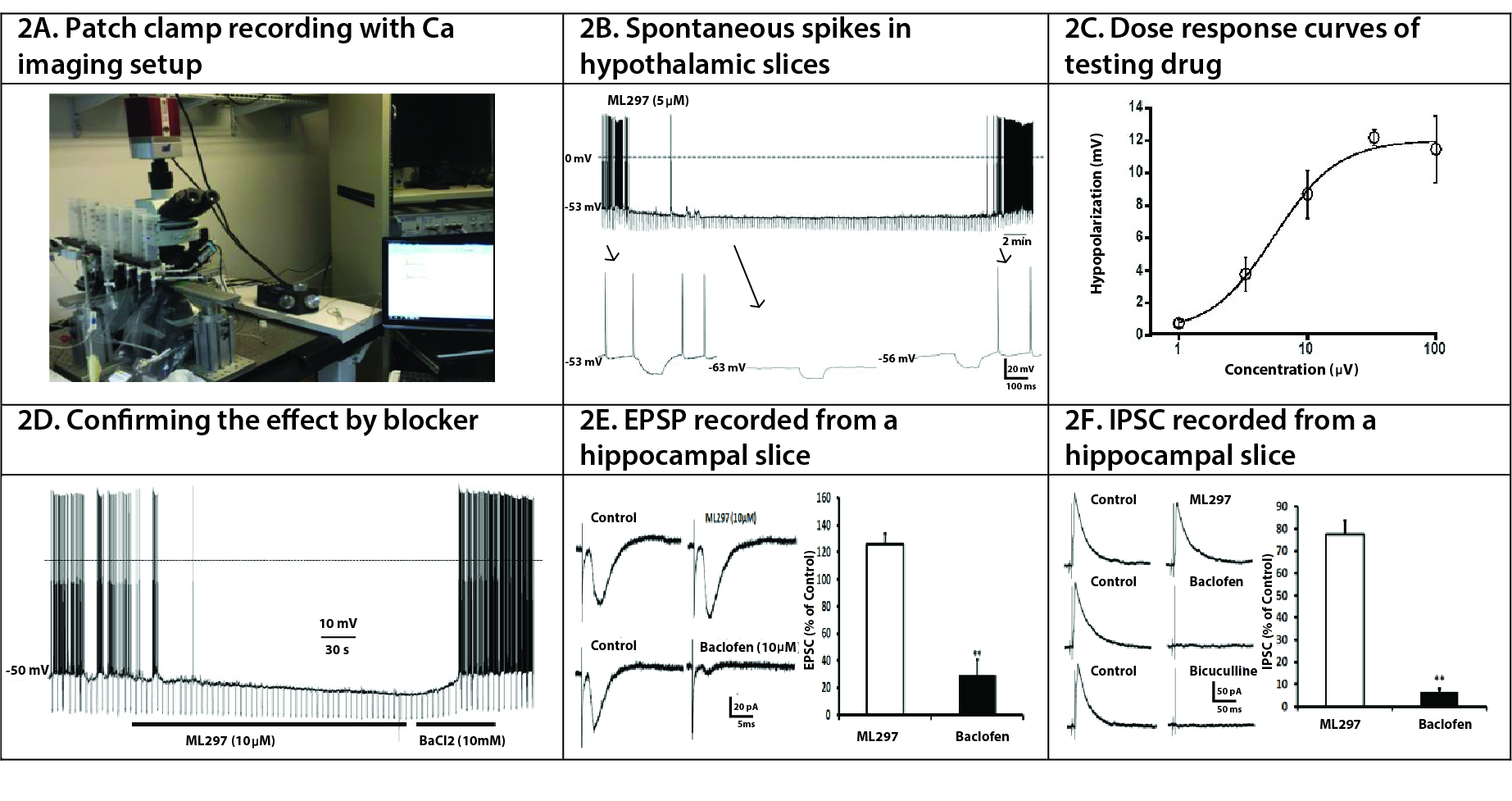
Figure 3. Mice were euthanized and the brains were quickly isolated and placed into ice-cold artificial cerebrospinal fluid (ACSF) continuously bubbled with 5% CO2/95% O2. The ACSF is composed of (in mM) NaCl 124.0, KCl 2.5, KH2PO4 1.2, CaCl2 2.4, MgSO4 1.3, NaHCO3 26.0 and glucose 10.0 (pH 7.4). Slices (260 μm thick) containing hippocampus and/or thalamus were prepared using a vibratome (Leica) and incubated at room temperature in continuously oxygenated ACSF for at least 1 h before recordings at room temperature. Slices were continuously perfused with ACSF bubbled with 5% CO2/95% O2 at a flow rate of 1 mL/min from an elevated reservoir. Recordings were made using an Axon 700B patch clamp amplifier, Axonpatch 1D for extracellular recordings, or a 64-channel multi-electrode array (MEA-64, Multi-Channel Systems).
Humanized Mouse Models in Immuno-Oncology Studies -
Human Immune System Reconstruction Mouse Models
4. Efficacy study in PBMC-engrafted M-NSG mice
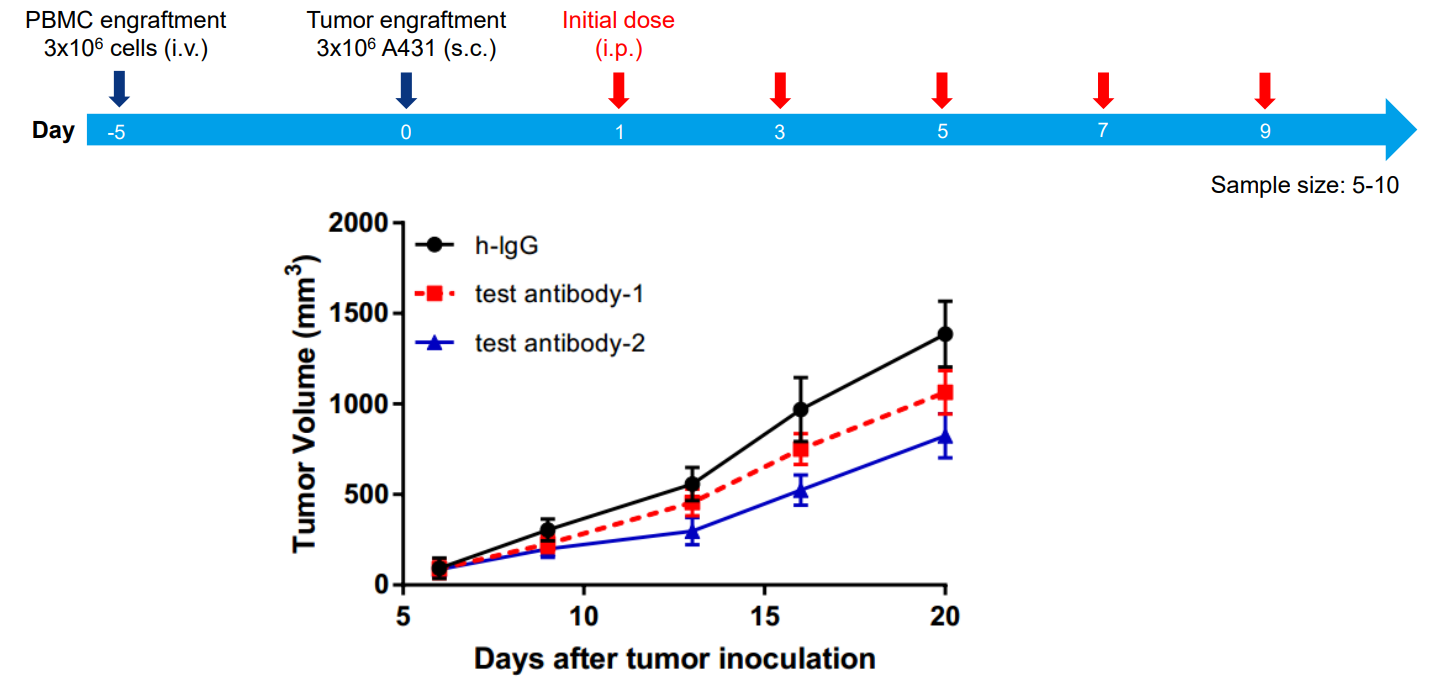
Figure 4: Tumor Volume - PBMC-engrafted M-NSG Mice Efficacy Study
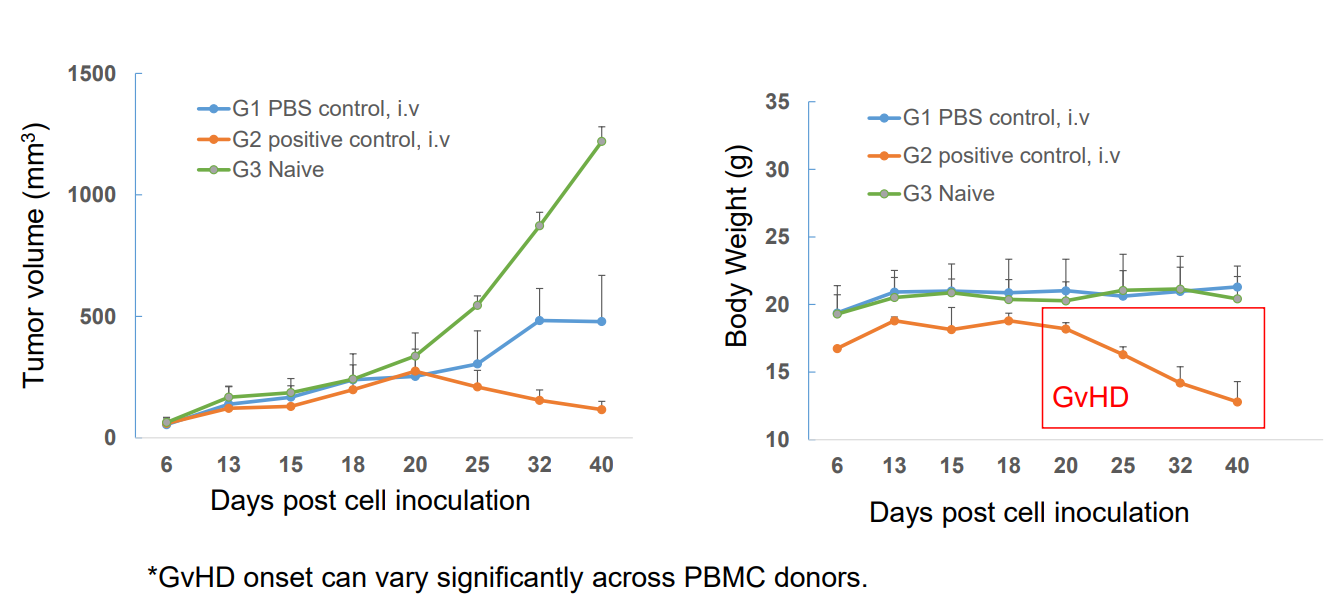
Figure 5: Tumor Volume (left) and Body Weight (right) - PBMC-engrafted M-NSG Mice Efficacy Study
Humanized Mouse Models in Immuno-Oncology Studies -
Immune Checkpoint Humanized Mouse Models
5. Humanized hcTLA4 Mouse
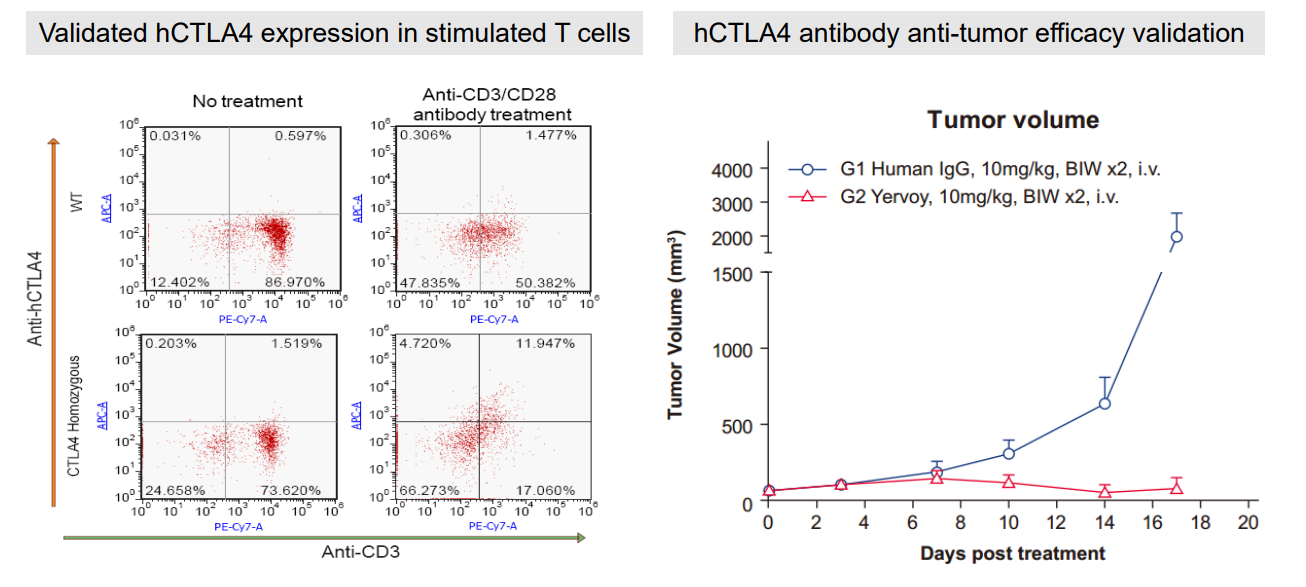
Figure 6: Validation of hcTLA4 expression in T cells (left) and hCTLA4 antibody anti-tumor efficacy validation (right).
6. Humanized OX40 Mouse
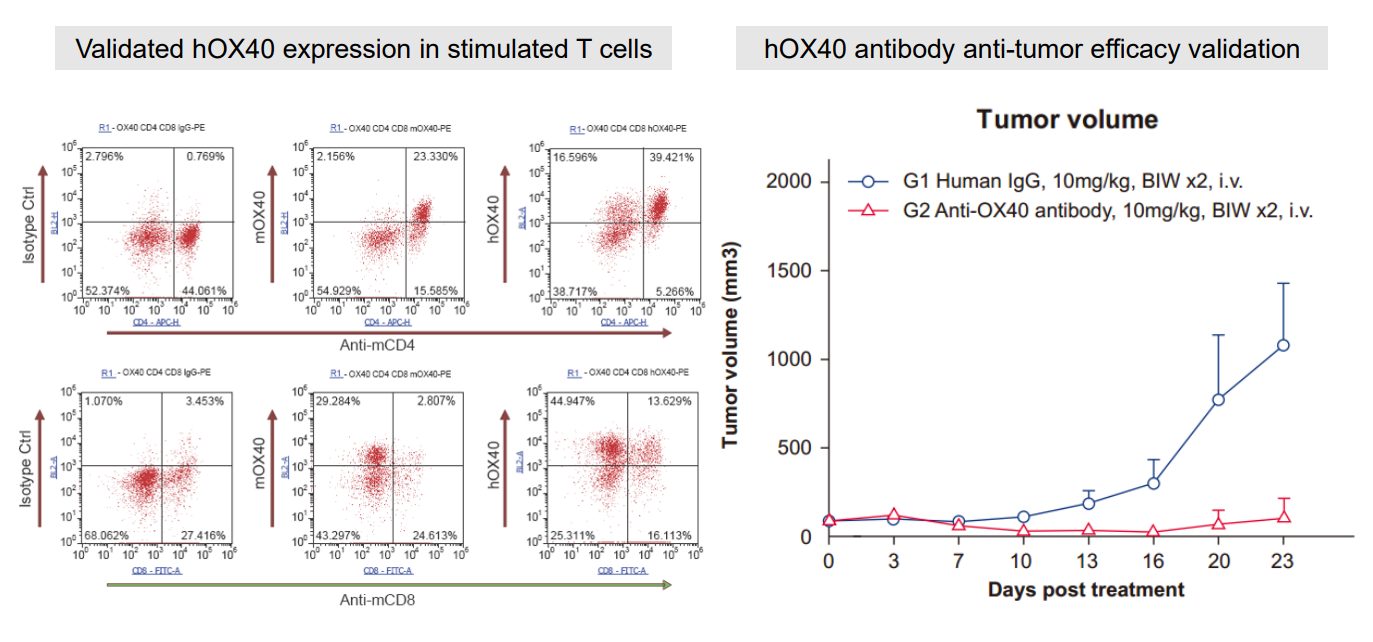
Figure 7: Validation of hOX40 expression in T cells (left) and hOX40 antibody anti-tumor efficacy validation (right).
7. Humanized LAG3 Mouse
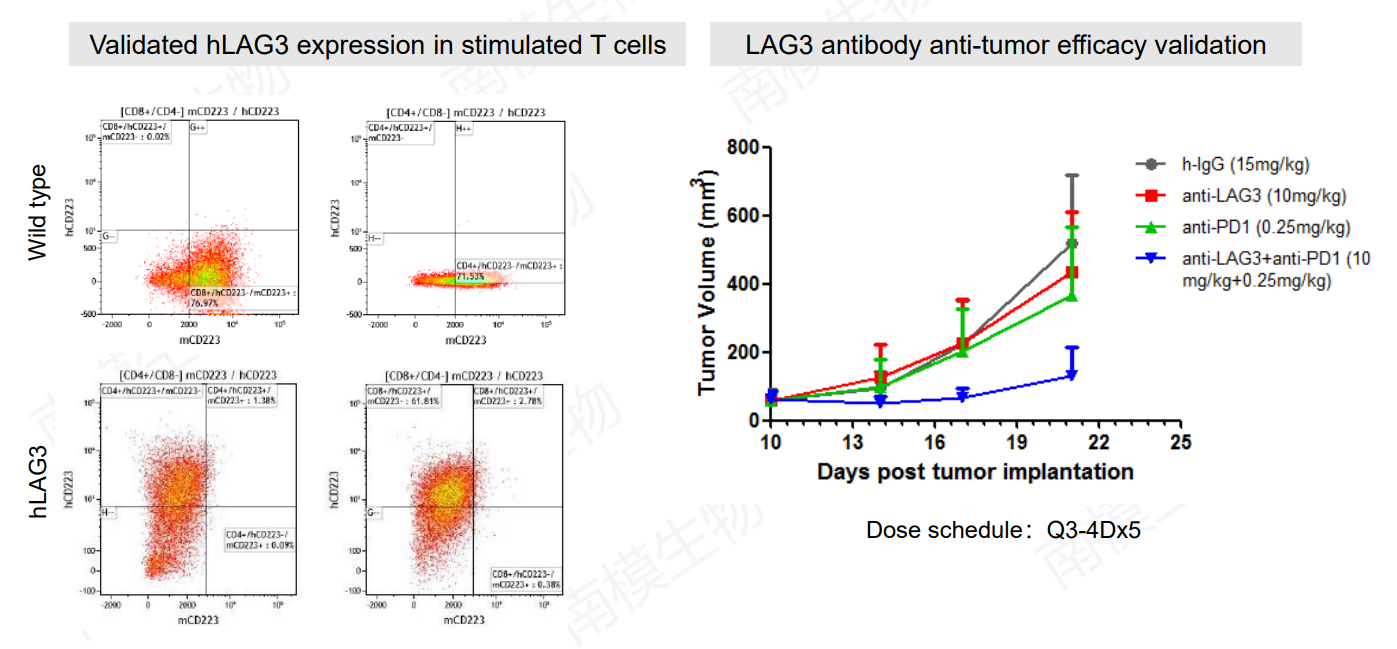
Figure 8: Validation of hLAG3 expression in T cells (left) and hLAG3 antibody anti-tumor efficacy validation (right).



 Knock-in/out, Conditional Knockout Mouse / Rat Model
Knock-in/out, Conditional Knockout Mouse / Rat Model