Newsletter
Neural Stem Cell & Neural Progenitor Cell Differentiation
The process of iPSC differentiation to neurons and neuronal cells is of special importance for neurobiology and related disorders, considering the dearth of clinically relevant in vitro models available for research, drug screening, and development, as well as the lack of therapy to reverse neuronal damage.
The true potential of iPSCs and ESCs rest in their directed differentiation to different somatic lineages. As part of our comprehensive stem cell service platform, researchers can leverage our expertise in stem cell technologies and our cost-effective, reliable iPS cell differentiation services for their developmental biology, disease mechanisms, and drug discovery research. Differentiate your patient-derived iPSCs into multipotent neural stem cells (NSCs).
- High differentiation efficiency and cell purity
- Our team can differentiate your iPSCs, one of ASC's control lines, or we can generate your iPSCs
- Cells are fully characterized for NSC biomarkers (i.e. SOX1 & Pax6) prior to delivery
- NPC Biomarker Characterization: Nestin
- Fast turnaround time
- NPCs - 3 weeks
- Inquire about iPSC differentiation to Neural Progenitor Cells
- GMP iPSC Differentiation Services Available >> Learn More
Also, avail our upstream iPSC generation and downstream cell-based assays services to analyze your differentiated cells or for drug screening applications.
Products and Services
Case Studies
Case Study 1
iPSC Differentiation into Neural Stem Cells (NSC)
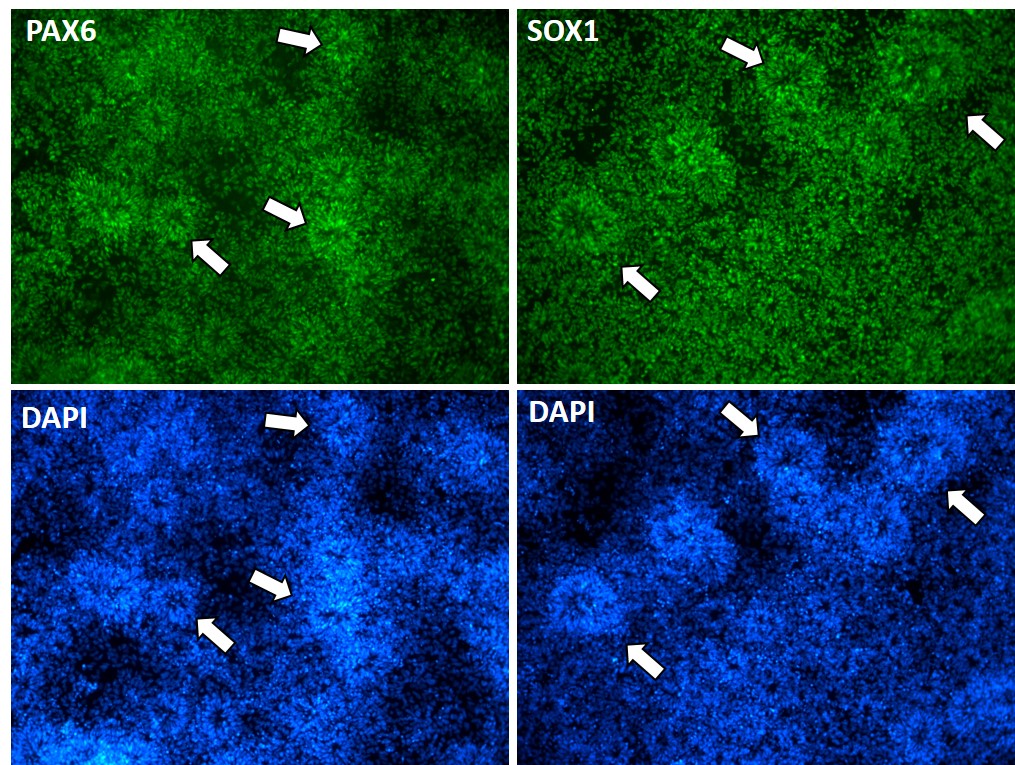
Figure 1. iPSC-derived neural stem cells (NSC) expressing key NSC markers PAX6 and SOX 1 (green) cultured in feeder-free condition, differentiated using Applied StemCell's proprietary neural induction protocol. DAPI: nucleus staining (blue); White arrows: neuronal rosettes.
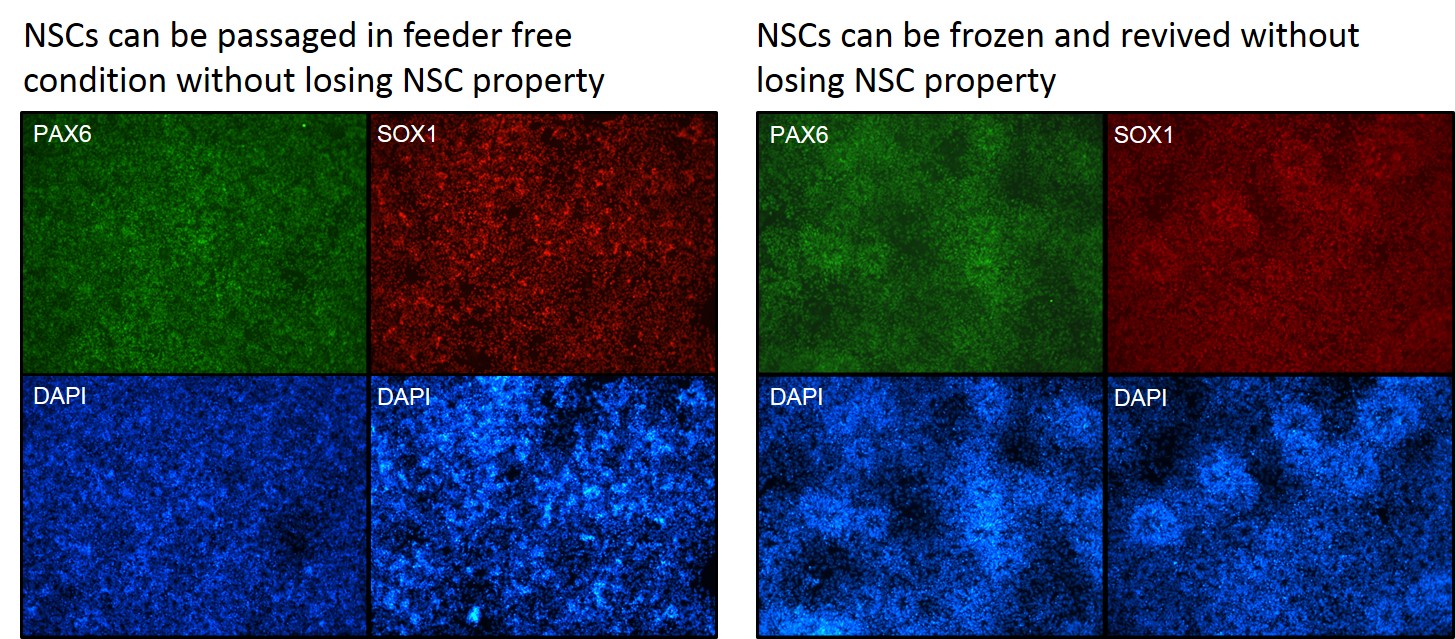
Figure 2. Differentiated NSCs retain neural stem cell phenotype even after free-thaw and passage under feeder-free condtions.




