Newsletter
Normal, Human Induced Pluripotent Stem Cells (hiPSCs)
iPSC Control Lines from Healthy Donors
Applied StemCell offers well-characterized, normal human induced pluripotent stem cells (iPSC) that have been reprogrammed from healthy human tissues (e.g., cord blood, fibroblasts, PBMCs) using footprint-free methods and Yamanaka factors. All iPSC lines have been carefully selected for morphology of undifferentiated cells, expression of pluripotency markers (OCT4, SOX2, SSEA4, and others), and strong endogenous alkaline phosphatase activity. Our normal human iPSCs have proven genome editing and differentiation capabilities, and are ideal for disease modeling, drug discovery/screening, and neurotoxicity screening applications.
We offer differentiated cell products from ASE-9211, including NSCs, astrocytes, dopa neurons, microglia, myoblasts, HPCs CD34+, NK cells, CD4/CD8 T cells, RPE, photoreceptors, cardiomyocytes, and more.
GMP iPSC Products & Services >> Learn More
iPSC Differentiated Cells >> Learn More
Products and Services
Application Notes
iPSC Differentiation (ASE-9211)
The control iPSC line, ASE-9211 was used to produce natural killer (NK) cells, T cells, and motor neurons using proprietary, integration-free differentiation protocols.
Natural Killer Cell Differentiation
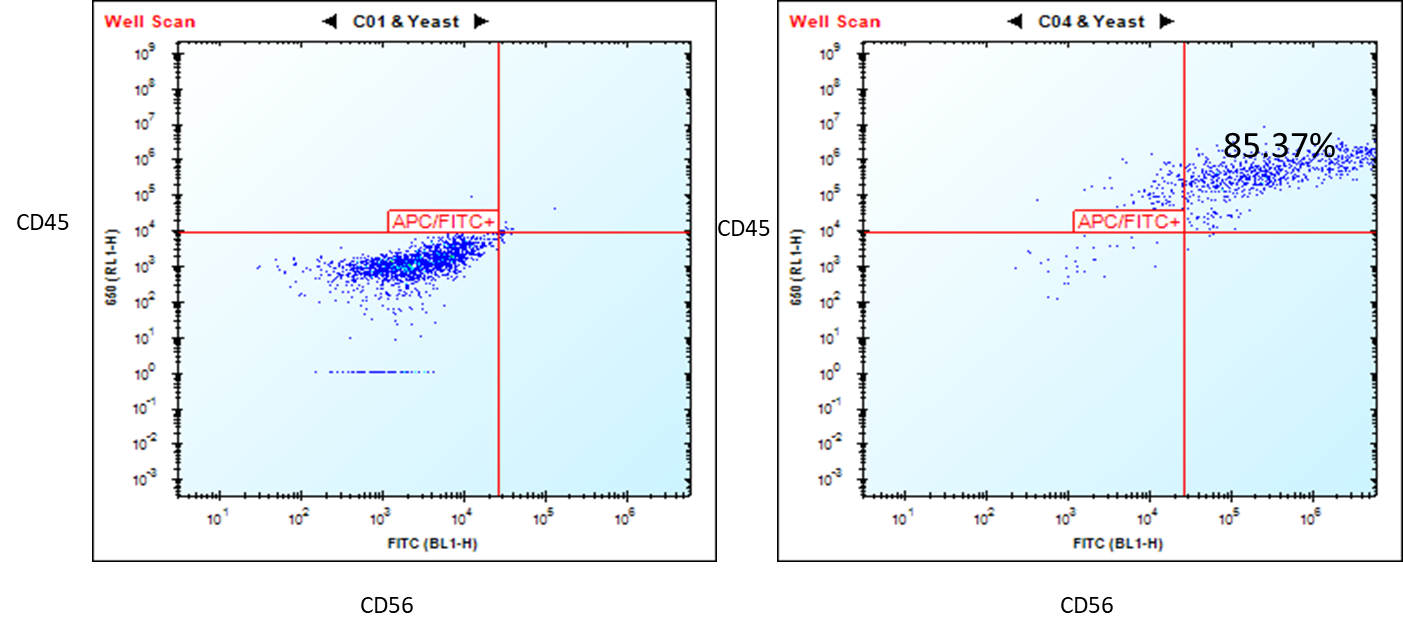
Figure 1. Flow cytometry analysis of ASE-9708 iPSC-derived NK cells for NK cell biomarkers. Cryopreserved NKs, differentiated from Applied StemCell’s control iPSC line, ASE-9211 were recovered in NK culture media. The cells were stained with NK cell markers, CD45 and CD56 at day 2. Left: Isotype control antibodies. Right: CD45/CD56.
T Cell Differentiation
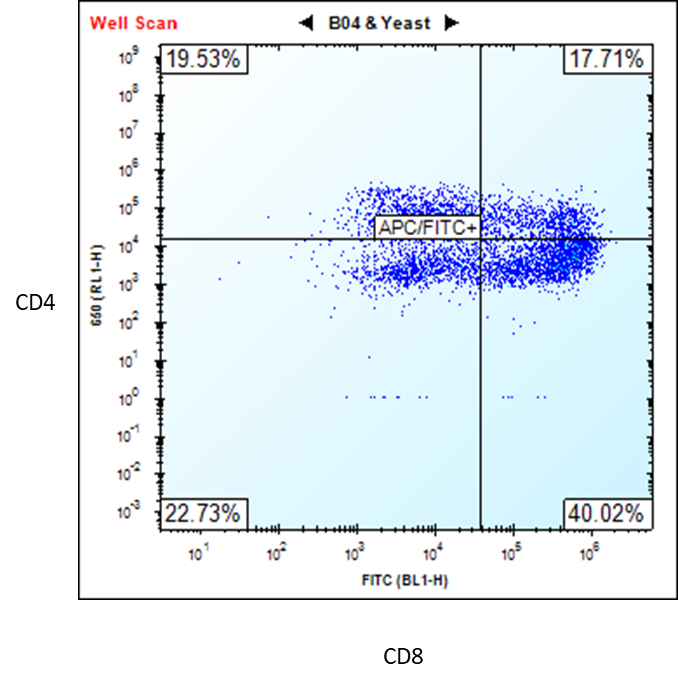
Figure 2. Flow cytometry analysis of ASE-9720 iPSC-derived T cells for T cell biomarkers. The T cells,
differentiated from Applied StemCell’s control iPSC line, ASE-9211, were stained with T cell markers CD4 and
CD8. (Premature T Cells CD4+/CD8+ = 17.71% and Mature T Cells CD8+ = 40.02%) *
*Upon request, the T cells can be purified by FACS or MACS (single positive or double positive). We can also further differentiate the cells to obtain a higher percentage (≥80) of mature CD8+ T cells.
Motor Neuron Differentiation
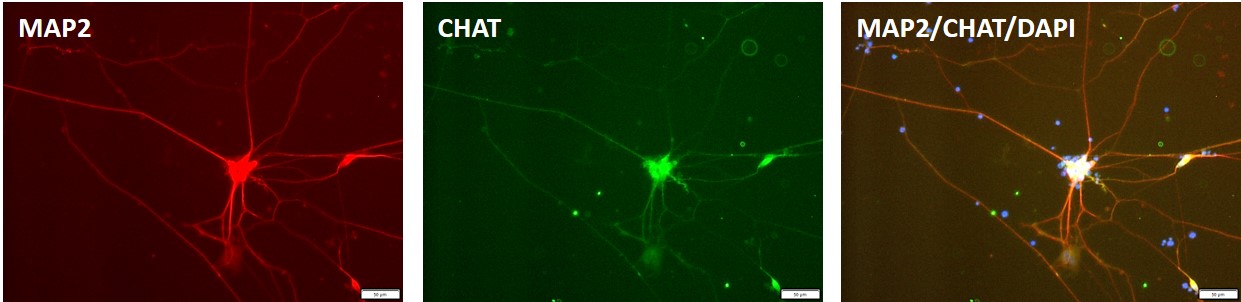
Figure 3. Immunocytochemical staining for motor neuron marker, CHAT (green) and the neuronal marker, MAP2 (blue) in motor neurons differentiated from control "master" iPSC line, ASE-9211 at 7 days post-thaw. DAPI (blue) was used for nucleus staining.
Publications
Control iPSC Lines:
- Tanaka, H., Homma, H., Fujita, K., Kondo, K., Yamada, S., Jin, X., ... & Atsuta, N. (2020). YAP-dependent necrosis occurs in early stages of Alzheimer’s disease and regulates mouse model pathology. Nature Communications, 11(1), 1-22.
- Su, S., Guntur, A. R., Nguyen, D. C., Fakory, S. S., Doucette, C. C., Leech, C., ... & Sims-Lucas, S. (2018). A renewable source of human beige adipocytes for development of therapies to treat metabolic syndrome. Cell reports, 25(11), 3215-3228.
- Lizarraga, S. B., Maguire, A. M., Ma, L., van Dyck, L. I., Wu, Q., Nagda, D., ... & Cowen, M. H. (2018). Human neurons from Christianson syndrome iPSCs reveal allele-specific responses to rescue strategies. bioRxiv, 444232.
- Tanaka, H., Kondo, K., Chen, X., Homma, H., Tagawa, K., Kerever, A., ... & Fujita, K. (2018). The intellectual disability gene PQBP1 rescues Alzheimer’s disease pathology. Molecular Psychiatry, 1.
- Kavyasudha C., Macrin D., ArulJothi K.N., Joseph J.P., Harishankar M.K., Devi A. (2018) Clinical Applications of Induced Pluripotent Stem Cells – Stato Attuale. In: Advances in Experimental Medicine and Biology. Springer, New York, NY. https://doi.org/10.1007/5584_2018_173.
- Lin, Y., Linask, K. L., Mallon, B., Johnson, K., Klein, M., Beers, J., ... & Zou, J. (2017). Heparin Promotes Cardiac Differentiation of Human Pluripotent Stem Cells in Chemically Defined Albumin‐Free Medium, Enabling Consistent Manufacture of Cardiomyocytes. Stem cells translational medicine, 6(2), 527-538.
FAQs
What is donor information for the ASE-9101 (iPSC from fibroblast, Male, Retrovirus)?
ASE-9211: Is it possible to thaw them on a Vitronectin XF ECM, or on an hESC-qualified Geltrex basement matrix?