Newsletter
iPSC Differentiated Microglia
Microglia play a crucial role in maintaining neuronal homeostasis and synaptic plasticity for normal brain development and neuronal function, and inflammatory responses. They have also been implicated in the pathogenesis of several neurological disorders such as Alzheimer’s disease and Parkinson’s disease. The differentiation of iPSCs to microglia provides a steady source of primary microglia-like cells without the sourcing issues associated with primary microglia. These cells are an excellent physiologically relevant research model to study immune response mechanisms in the brain, neuronal function, and disease modeling.
Applied StemCell (ASC) offers high-quality iPSC-derived, cryopreserved microglia cells that have been differentiated using a proprietary, integration-free method. The differentiation mimics the in vivo erythromyeloid induction and activation pathway for the development of microglia in the brain to generate physiologically relevant cell lines. These microglia-like cells have been derived from a well-characterized control human iPSC line (ASE-9211), and express microglial-specific markers, P2RY12 and TMEM119 as well as macrophage markers. These are functionally active, high-purity (>90%) microglial cells that are ideal as controls for disease modeling, drug screening, and co-culture modeling. Also, included with the microglia we also provide proprietary media to ensure robust microglial cultures.
- High-purity: >90%
- Microglial-specific markers: P2RY12 and TMEM119
- Integration-free differentiation
- GMP iPSC Products & Services >> Learn More
We also supply several other well-characterized iPSC-derived products, including Hematopoietic Progenitor Cells. With our comprehensive iPSC platform, we can even generate iPSCs or help you develop any downstream assay you may need.
Products and Services
Case Studies
Case Study 1
iPSC-derived Microglia (ASE-9601) from Control iPSC Line ASE-9211
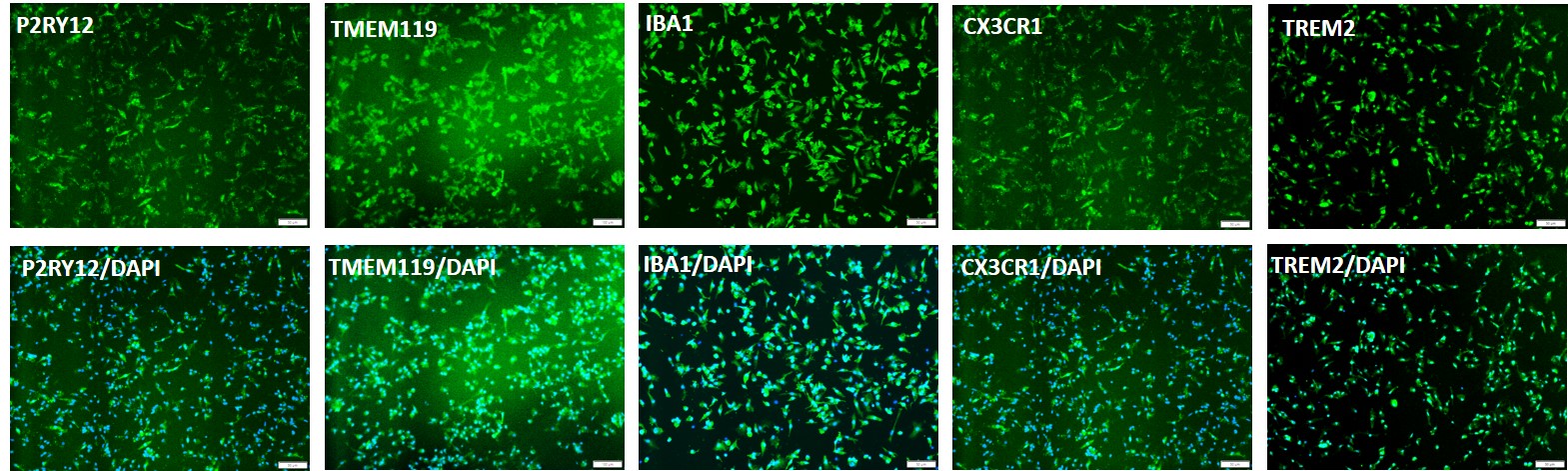
Figure 1. Cryopreserved iPSC-derived microglia (iMGLs; ASE-9601). Cryopreserved iMGLs differentiated from Applied StemCell’s control “master” iPSC line, ASE-9211 were recovered in microglia culture media. The cells were fixed the next day and stained with microglial-specific markers, P2RY12+, TMEM119, IBA1, CX3CR1, TREM2 (top row). Bottom row shows the co-localization of the biomarkers with the nuclear counterstain, DAPI.
FAQs
Can the iPSC-derived microglia be passaged?
Is the media provided serum-free?
Do I need to add any additional growth factors to maintain the microglia culture?
Which differentiation protocol was this line following?
How long does it take the cells to fully differentiate/mature?
Do you recommend a medium change (complete or partial) the day after plating? (ASE-9601)
From the datasheet, it seems that these cells can be grown and amplified, within a certain period of time. Is this really the case? If so, do you recommend culture conditions (flask sizes and associated cell density, subculture frequency, ...)?