Newsletter
iPSC Differentiated Neural Progenitor Cells
Neural Stem Cells (NSCs) are multipotent cells derived from iPSCs and ESCs that are self-renewing and have the potential to differentiate into various neuronal lineages. This makes them very attractive for in vitro patient-specific neuroscience research, whereby NSCs can be derived from patient-specific iPSCs and further differentiated into neurons and astrocytes.
Applied StemCell offers high-quality neural stem cells and neural progenitor cells (NPCs) derived from fully characterized iPSC line, ASE-9211. These NSCs & NPCs have been derived using integration-free protocols and express relevant biomarkers. The NSCs/NPCs retain their multipotency and neural marker even after cryopreservation and passaging. These fully characterized cells at low passage can be further differentiated, thus facilitating studies in cell replacement therapies and neuronal disease modeling.
Advantages of choosing ASC’s iPSC-derived neural stem cells/neural progenitor cells:
- High purity cells (≥95%) expressing neural stem cell marker: Nestin (other markers available upon request)
- Fully characterized by immunocytochemistry and whole-genome profiling
- Provides a consistent and reliable source of differentiated mature, functional neurons and astrocytes
- Physiologically relevant cell line models
- GMP iPSC Products & Services >> Learn More
Custom Differentiation Service for iPSC-derived NSC: We offer direct differentiation of your control/patient/engineered iPSC lines into neural stem cells. Service includes full characterization of the derived cell line.
Products and Services
Case Studies
Case Study 1
Characterization of iPSC-derived NPCs (ASE-9740)
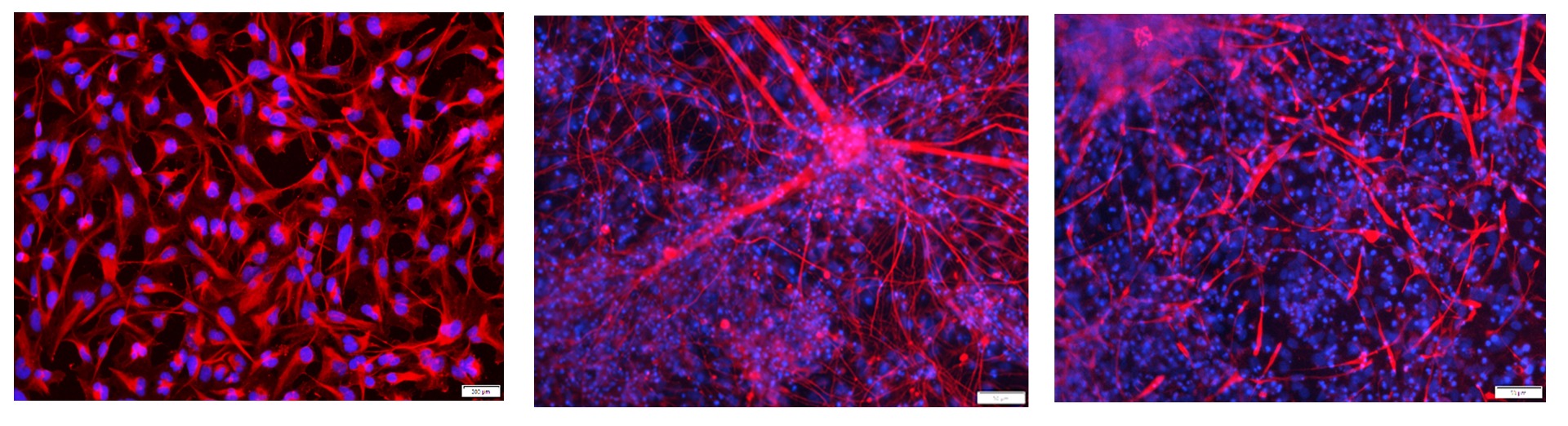
Figure 1. Immunostaining of the ASE-9740 Neural Progenitor Cells derived from Applied StemCell’s control iPSC line ASE-9211. The neural progenitor cells (NPCs) (Left, Red: Nestin; Blue: DAPI) can be further differentiated into neurons (Middle, Red: Tuj1; Blue: DAPI) and astrocytes (Right, Red: GFAP;
Blue: DAPI).
Application Notes
- Neural development
- Neurotoxicity
- Gene Profiling during differentiation
- Disease Modeling
- Electrophysiology
- Co-culture applications
Publications
iPSC-differentiated Cell Lines
- Gupta, G., Gliga, A., Hedberg, J., Serra, A., Greco, D., Odnevall Wallinder, I., & Fadeel, B. Cobalt nanoparticles trigger ferroptosis‐like cell death (oxytosis) in neuronal cells: Potential implications for neurodegenerative disease. The FASEB Journal.
- Shaltouki, A., Sivapatham, R., Pei, Y., Gerencser, A. A., Momčilović, O., Rao, M. S., & Zeng, X. (2015). Mitochondrial alterations by PARKIN in dopaminergic neurons using PARK2 patient-specific and PARK2 knockout isogenic iPSC lines. Stem cell reports, 4(5), 847-859.
- Efthymiou, A. G., Steiner, J., Pavan, W. J., Wincovitch, S., Larson, D. M., Porter, F. D., ... & Malik, N. (2015). Rescue of an in vitro neuron phenotype identified in Niemann-Pick disease, type C1 induced pluripotent stem cell-derived neurons by modulating the WNT pathway and calcium signaling. Stem cells translational medicine, 4(3), 230-238.
- Efthymiou, A., Shaltouki, A., Steiner, J. P., Jha, B., Heman-Ackah, S. M., Swistowski, A., ... & Malik, N. (2014). Functional screening assays with neurons generated from pluripotent stem cell–derived neural stem cells. Journal of biomolecular screening, 19(1), 32-43.
- Shaltouki, A., Peng, J., Liu, Q., Rao, M. S., & Zeng, X. (2013). Efficient generation of astrocytes from human pluripotent stem cells in defined conditions. Stem cells, 31(5), 941-952.
FAQs
Any other details you could please share on the NPCs?
Let's look at the leftmost illustration in the datasheet, ASE-9740: To generate this image, how much time (in days) passed between plating in 96 wells and fixing the cells? And what cell density was initially used?
Are the NPCs made through Embryoid body protocol or Monolayer protocol?
In the datasheet, it says at the end that "split the cells every 3-4 days. Does this mean that these cells can be maintained in culture for several passages? If so, how many passages?