Newsletter
Microglia Differentiation
Induced Pluripotent Stem Cells (iPSCs) are popular because their pluripotency allows them to differentiate into all three germ layers with the potential to differentiate into all cell types within the body, but unlike ESCs, they are not encumbered with the ethical dispute associated with the sourcing of ESCs. The true potential of stem cell technology lies in the directed terminal differentiation of iPSCs into specific somatic cells which function like primary cells but without the sourcing issue and passaging limitations associated with them. The directed differentiation of iPSCs to glial cells (microglia, astrocytes, and oligodendrocytes) and neuronal lineage cells (neural stem cells and neurons) is of special importance for neurobiology and related disorders, considering the dearth of clinically relevant in vitro models available for research, drug screening, and development, as well as the lack of effective therapy to reverse neuronal damage.
Differentiate your iPSCs into microglia, the resident immune cells of the central nervous system (CNS) for an efficient way to generate in vitro models of neurodevelopment, neuroinflammation, and neurological disorders such as Parkinson’s disease and Alzheimer’s disease. iPSC-derived microglial cells recapitulate the phenotypes and functional properties of primary microglial cells without the sourcing problems associated with them.
Advantages of ASC's Comprehensive iPSC Microglia Differentiation Standard & Custom Services:
- Optimized differentiation protocols
- Receive robust, mature microglia with the morphology of primary microglia
- Cells express key microglial specific markers: IBA1, TMEM119, P2RY12, CX3CR1, TREM2; other markers available upon request
- Differentiate from your healthy, disease or engineered iPSCs
- Control “Master” iPSCs and iPSC Generation are available for deriving your control microglial lines
- Fast turnaround time (2 months)
- GMP iPSC Differentiation Services Available >> Learn More
Optional! Add-on our downstream, cell line validation and phenotype assessment assays for a complete and comprehensive cell line package.
Products and Services
Application Notes
Case Study 1
iPSC-derived Microglia from Control iPSC Line ASE-9211
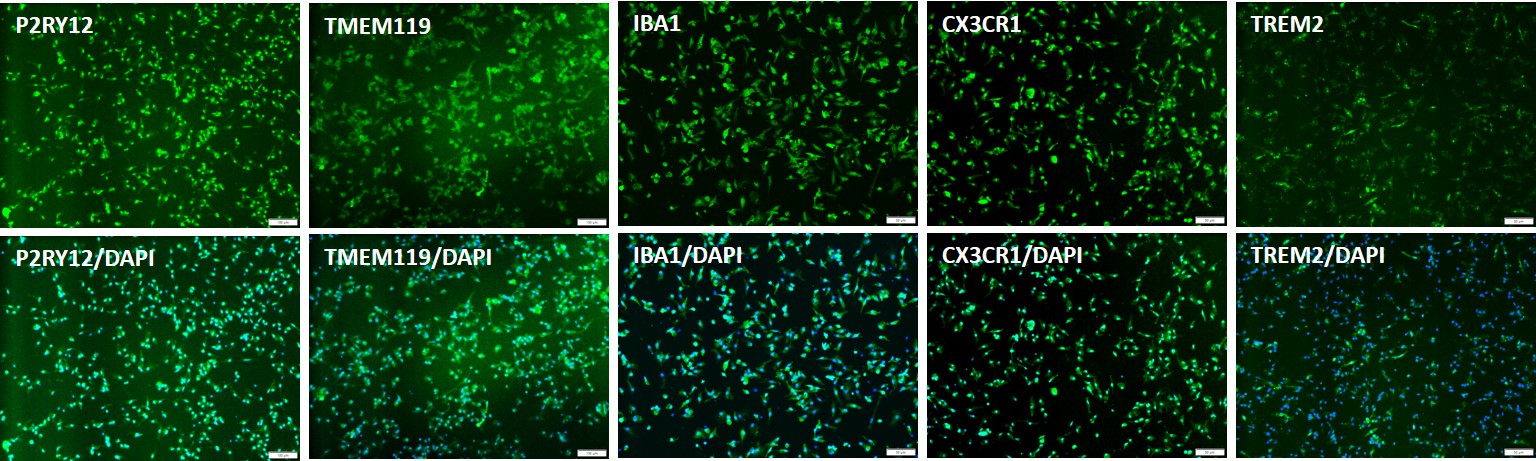
Figure 1. Recovery of cryopreserved iPSC-derived microglia (iMGLs). Cryopreserved iMGLs differentiated from Applied StemCell’s control iPSC line, ASE-9211 were recovered in microglia culture media on plates pre-coated with a proprietary microglia-specific coating matrix. The cells were fixed the next day and stained with microglial-specific markers, P2RY12+, TMEM119, IBA1, CX3CR1, TREM2 (top row). Bottom row shows the co-localization of the biomarkers with the nuclear counterstain, DAPI.
FAQs
Will the iPSC-derived microglia provided be precursors or mature cells?
What is the purity of the microglia?
What are the quality control criteria you would provide?
What is your differentiation protocol based upon?
Do you provide media and protocols along with the microglia?
Can the differentiated microglia be passaged?
How long can your differentiated microglia be maintained in culture?



