
Newsletter
Accelerate your research and discoveries by leveraging our extensive expertise in stem cell technology! We can provide physiologically relevant in vitro models derived from Pluripotent Stem Cells (PSC) for more predictive models of human disease and biology. ASC offers a comprehensive custom service platform for every aspect of stem cell-based research: iPSC generation/ ESC derivation, CRISPR/Cas9 genome editing and cell line model generation; differentiation to somatic lineage of choice, downstream assays to validate your cell line and for drug discovery and screening.


Applied StemCell has a comprehensive catalog of products for every aspect of your stem cell research:
ASC is a leading provider of genetically engineered mouse models for biomedical research and preclinical drug discovery. With 13+ years of expertise in mouse model engineering and >500 engineered mouse models under our belt, we can engineer advanced, physiologically relevant mouse models with a wide range of precision genetic modifications specific to your projects’ needs.
We also offer a repository of off-shelf mouse models that can be directly used for studies on gene function, drug screening, and human diseases, thus effectively shortening your overall experimental cycle.
Applied StemCell, a long-standing leader in custom mouse model engineering, also offers 2,000+ “off-shelf” proprietary, genetically engineered mouse models (GEMM) that are research-ready and can be directly used in gene function, drug screening, and human disease research.





Genetically engineered rat models are gaining popularity as the preferred biological model for several research areas. ASC can genetically engineer rat models with physiologically relevant modifications using an expanded technology portfolio with CRISPR/Cas9 and traditional homologous recombination and random transgenic technologies.
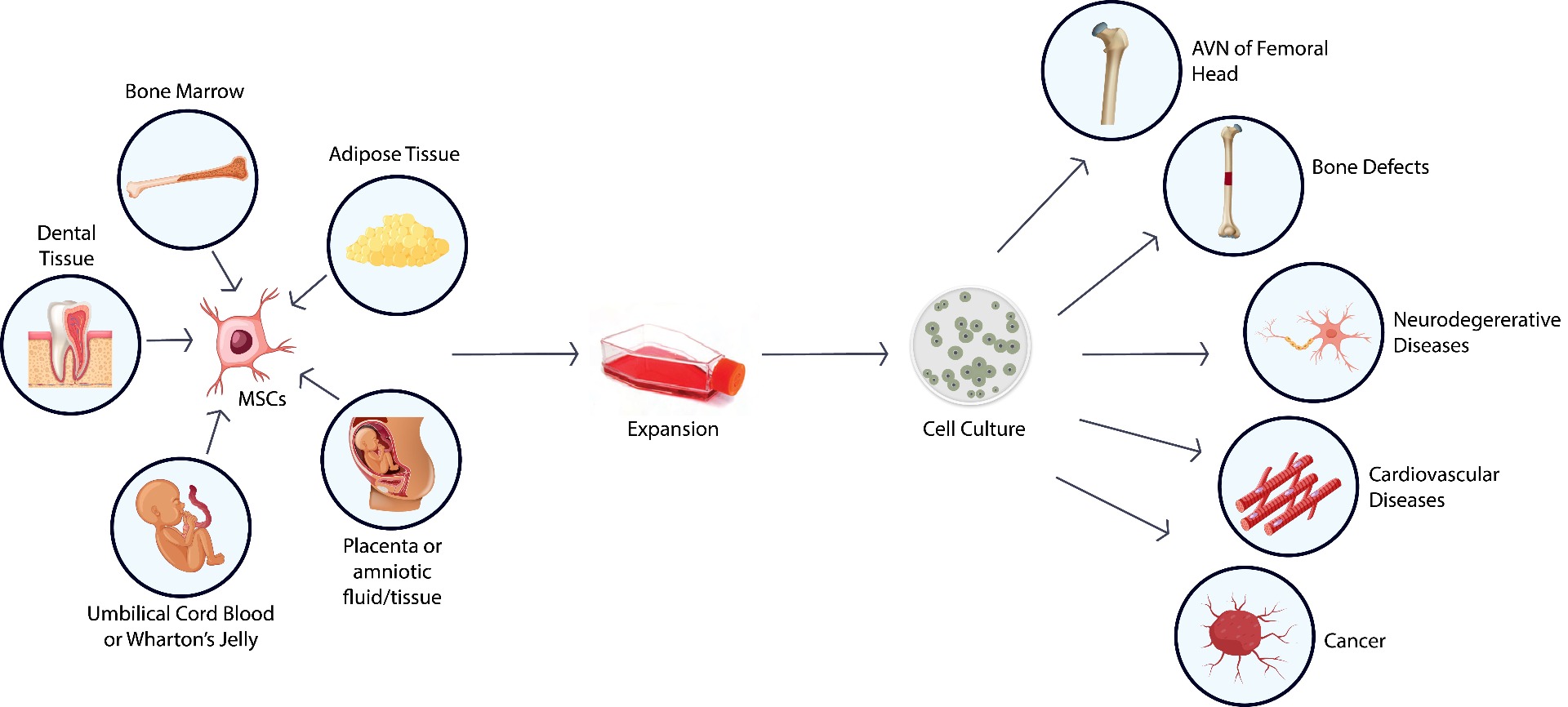
Mesenchymal stem cells (MSCs) have the capacity of multi-direction differentiation making them an ideal cell therapy starting material. At ASC, we offer end-to-end stem cell solutions, including cell isolation, expansion and banking, characterization, and differentiation.
End-to-end MSC solutions for cell therapy manufacturing
Applied StemCell’s proprietary site-specific, integrase-based TARGATT™ technology has been used to generate stable, high-quality TARGATT™ Master iPS, CHO, and HEK293 cell lines for large transgenes knock-in. However, if you would like to knock in your gene-of-interest (GOI) into your own cell line, our experts can engineer the TARGATT™ landing pad into your cells. With your engineered TARGATT™ cell line and our unique integrase, you can insert a single copy of your DNA fragment (up to 20kb) at a safe harbor locus. If you would like to learn more, contact us today to schedule your free consultation.
As a long-standing leader in genetically engineered animal models, Applied StemCell also offers a comprehensive custom research solutions platform for downstream assays in mouse and rat models. Our state-of-the-art vivarium is equipped with automated cages & devices for behavioral assessments, sample collection and in vivo measurements (ECG, EEG) of the animals. We have a multidisciplinary team of expert scientists who can design a comprehensive project plan to fit any requirement/stage of your research pipeline and for drug discovery and screening.
Cell based assays have become an integral part of preclinical studies to elucidate biological mechanisms, and for screening drugs for efficacy, toxicity or combination analysis for drug synergy or antagonism. The use of live cells in these assays provide a fast and biologically relevant source of information. Applied StemCell offers a fully comprehensive panel of Cell-Based Assays from cell line expansion, cell line model generation, validation, to assays that evaluate cell line function and behavior. We have extensive experience in culturing and handling a wide variety of cell lines including cancer cell lines, immortalized cell lines, stem cells and differentiated cell lines. We will work with you at every stage of your project to fit it to the exact needs of your research.
A comprehensive custom Lentiviral-based Stable Cell Line Generation Service for genetically engineering sensitive cells, primary cells, stem cells and terminally differentiated cells. Lentivirus-based stable cell line generation offers a cost-effective and efficient alternative for generating stable cell line models for gene overexpression, inducible gene expression, gene knockout and gene knockdown.
Key Features:
With >10 years’ experience in molecular biology and genome editing technologies, ASC offers start-to-finish customized services for DNA cloning. vector design and lentivirus/ retrovirus packaging. We can develop the best strategies to tackle your cloning projects and technical problems.
.jpg)

 Design and Cloning.jpg)
Applied StemCell offers gamma-irradiation service for irradiating cell lines (patient-derived, pooled, genome edited/ corrected cell lines) using a Nuclear Regulatory Commission (NRC)-approved Gamma-emission source to irradiate cells at a constant dose rate and to any desired extent of radiation. Irradiated cell line models are novel toolkits for applications such as generation of co-culture models using non-dividing/ feeder cells; to study mechanisms underlying cancer progression; wound healing and inflammatory responses after irradiation; screen drugs for enhancing irradiation sensitivity of tumor cells; and for xenograft tumor models.
Our Cell Line Irradiation Service includes:
Primary cells are isolated from tissues, including blood and bone marrow. The isolated human primary cells have the capacity to function similarly to cells in vivo under the right conditions, and they can be incorporated into various areas of research as model systems. Primary cells can be used for drug discovery, toxicity testing, gene therapy, cancer research, and more.
In order to address the needs of the research community, Applied StemCell (ASC) now offers high-quality fresh and frozen human primary cells, including peripheral blood cells, cord blood cells, and bone marrow cells. ASC also provides hematopoietic stem cells, progenitor cells, and immune cells. For fresh cells, we offer on-demand, customized sample collection and processing services. All donors are IRB-approved and have been tested for Syphilis, Hepatitis B (HBV), Hepatitis C (HCV), HTLV-I/II, HIV-1, and HIV-2. Same-day delivery is available for Bay Area customers. Overnight delivery and international shipping are available for all other customers. Contact us today to learn more.
 Sample Human Primary Cell Datasheet
Sample Human Primary Cell Datasheet



Applied StemCell (ASC) now offers human hematologic tissue products, including human healthy whole/peripheral blood and cord blood samples. Plasma, Leukopak, and Off-the-clot serum products are available. ASC offers on-site procurement and processing. Our samples are collected from IRB-approved donors who have tested negative for Syphilis, Hepatitis B (HBV), Hepatitis C (HCV), HTLV-I/II, HIV-1, and HIV-2. Contact ASC today to learn more about customized sample collection and requesting a specific donor.




Non-human primate (NHP) cells and hematologic products are now available. Our non-human primate samples are collected from healthy cynomolgus macaques or rhesus macaques that are housed in the United States. All of our products are from NHPs that have tested negative for SIV, STLV, SRV, and Herpes B. These high-quality NHP products can be used in disease modeling, toxicity testing, or other areas of research. You can schedule a free consultation today to discuss specific project requirements with one of our experts. You can request an NHP donor of a specific gender or age, and you can reserve the cynomolgus or rhesus donor for the entire duration of your project. Contact us today to learn more.




Applied StemCell (ASC) works closely with clinicians to obtain disease samples from patients who have been recently diagnosed or have not received treatment. ASC provides a diverse selection of human fresh and frozen diseased products that can be used for disease modeling, drug development, and other areas of research. Call us today to place your order. We will have the sample ready and, in your hands, the same day of collection when you order one week in advance. ASC offers same-day delivery to Bay Area customers, and overnight and international shipping is available to all customers. The availability of the diseased products is dependent on the prevalence of the disease. If you would like more information, contact us today!




Applied StemCell (ASC) now offers high purity cytokines and growth factors produced by HEK293T cells. We use novel technology to provide customers with high-purity human cytokines that have not been tagged during production. Additionally, our extensive purification methods allow us to generate human cytokines that exhibit super-low levels of endotoxins. ASC’s high purity and high activity cytokines allow you to avoid the higher levels of endotoxin contamination of cytokines from bacteria and other systems. Contact ASC today to speak with one of our experts to learn more about our active human-specific cytokines and which product best fits your project needs.




If you are looking to expedite your project timeline and you do not require customized FFPE slides, scrolls, or blocks, explore our new collection of FFPE cell pellets and tissues to discover the high-quality FFPE product that can help you drive your research forward. Applied StemCell provides FFPE human and animal cell pellets in various formats, including slides, blocks, and scrolls. Our technology enables the production of round, homogenous pellets that are evenly distributed throughout each section. Although we supply a diverse set of cell pellets with consistent densities, FFPE human and animal tissue products are also available for purchase. When you find what you are looking for, contact us and we will help you place your order.

 Sample FFPE Cell & Tissue Datasheet
Sample FFPE Cell & Tissue Datasheet| Breast | Lung | Cervical | Prostate |
|
| SKBR3 MCF7 MDA-MB-231 MDA-MB-468 3 BT474 T47D MDA-MB-453 Hs578T BT549 ZR751 BT20 MDA-MB-175 HCC1806 HCC1143 HCC70 HCC1937 HCC38 DU4475 |
H2228 H23 A549 HOP62 H322 H1703 H1975 H838 SK-MES-1 HOP92 H596 H1650 SW1573 HCC78 MRC5 EKVX SW900 H226 |
H522 H460 DV90 H520 H2122 H647 H211 H820 H1755 H441 H358 H1299 H1993 H1648 HCC827 H841 HCC827GR6 |
HELA CASKI SIHA C33A HT3 MS751 ME180 |
PC3 DU145 22RV1 LNCAP |
| Colorectal | Skin |
| SW620 HT29 LS174T COLO205 HCT116 HCT15 SNU-C2B SNU-C1 SW48 SW1116 SW403 CACO2 Colo201 SW1417 SW948 LOVO SW480 |
SK-MEL-2 M14 CHL1 SK-MEL-28 C32 HT144 MALME-3M UACC62 SK-MEL-5 A431 HMCB SK-MEL-30 OPM-2 A375 Detroit551 BJ-5TA |
| Leukemia | Lymphoma | Ovarian | Kidney |
| K562 JURKAT HL60 CCRF-CEM MOLT4 RPMI-8226 THP1 THP1 MEC1 |
SUDHL1 SUDHL4 HUT78 SUDHL6 RI-1 WSU-NHL RAMOS MM1S DAUDI ST486 U937 HDLM-2 U266 FARAGE SR RAJI MV411 Pfeiffer |
OVCAR8 IGROV1 PA1 CAOV3 SKOV3 SW626 OVCAR3 OVCAR3 |
786-O HEK293 293T A498 CAKI-1 ACHN A704 769P |
| Liver | Pancreatic | Brain | Stomach | Head & Neck | Uterus | Bladder | Vulva |
| HEPG2 HUH7 HEP3B SNU475 |
BXPC3 MIAPACA2 PANC1 PSN1 PL45 ASPC1 CAPAN2 SW1990 |
U118MG A172 PFSK1 SW1088 DAOY U87MG U251MG SF295 SF295 DBTRG-05MG |
SNU16 SNU16 KATOIII Hs746T N87 SNU5 |
DETROIT-562 FADU SCC90 SCC152 SCC25 |
AN3CA | RT4 SW780 HT1197 |
SKLMS1 |
| Bone | Thyroid | Retroperitoneum | Muscle | Esophagus | Placenta |
| U2OS SH-SY5Y MG63 KG1 (CCL-246) SJSA1 |
Thyroid | SKPNDW 93T449 |
A673 | OE19 KYSE70 |
JEG3 |
| Animal | |
| 3T3 CHO CHO-K1 CHO-K1 NRK-49F NRK-49F aTC1 clone 6 EML beta-TC-6 MC/9 NE-4C B16-F10 C8-D1A CW13.20-3B3 |
CW13.20-3B3 EMT6 AML12 LL/2 (LLC1) N1E-115 CATH.a BW5147.3 MC38 Raw 264.7 bEND.3 Neuro2a CT26 |
| CPA | Cell Lines Included |
| Breast | MCF7, BT474, SKBR3, MDA-MB-468, MDA-MB-231 |
| Lung | A549, Hcc78, H1975, SKMES1, H1703, H596, HOP62, HOP92 |
| Prostate | PC3, DU145, 22RV1, LNCAP |
| Colorectal | SW620, LS174T, COLO205, HT29, HCT116, HCT15 |
| Leukemia/Lymphoma | JURKAT, CCRF-CEM, K562, RAJI, HUT78, SUDHL1, SUDHL4 |
| Pancreatic | PANC1, MIAPACA2, BXPC3, PSN1 |
| Skin | CHL1, C32, M14, SKMEL2, SKMEL28 |
| HER2 | HER2-0+, HER2-1+, HER2-2+, HER2-3+ |
| PD-L1 | PD-L1-, PD-L1+ |
Dont's see what you are looking for? Contact us today!
Applied StemCell can isolate primary cells from fresh tissues, including from biopsy samples.
Whether you are looking for formalin fixed paraffin embedded (FFPE) blocks, slides, scrolls, or arrays, we can generate the preserved human or animal product you need for your basic research, disease studies, or drug development project with our collection of customizable cell pellet and tissue FFPE services. Unlike other providers, our unique protocols enable the creation of round cell pellets with a consistent density in every section. If you are looking to preserve a specific animal tissue, our scientists can not only tailor your array to your core size and orientation requirements, our team can combine your cell pellet and tissue samples into a single array. Furthermore, you have the power to further personalize your service by selecting a different cell pellet shape (square, rectangle, etc.) and density (1-90%) or tissue thickness and length. If you would like to learn more, visit our service pages or contact us to schedule your free consultation.
ASC Service Advantages




TARGATT™ technology enables the site-specific integration of any gene-of-interest (GOI; up to 20kb) at a preselected safe harbor locus with the use of a unique integrase. Applied StemCell has generated many TARGATT™ cell lines, including the TARGATT™ CHO and HEK293 Master Cell Lines. These high-quality cells are now part of ready-to-use TARGATT™ knock-in kits that you can use to insert your GOI in your own lab. We have observed steady, medium to high levels of protein expression when using our master cell lines for customer projects, so you can confidently purchase any kit that best suits your research needs. Additionally, our experts have used engineered TARGATT™ cells to address common bioproduction issues.
The traditional CHO antibody and animal bioproduction methods are inefficient because they require random gene insertion and forced amplification of transgenes. Applied StemCell has addressed these problems by utilizing our expertise in cell line and animal model engineering with our proprietary TARGATT™ technology to develop two bioproduction methods: TARGATT™ CHO Master Cell Line and Transgenic TARGATT™ Rabbits. Both methods offer low cost, efficiency, consistent protein expression and high protein yield that is easily scalable for large-scale bioproduction.
Advantages of the TARGATT™ Master Cell Line:
If you would like to work with your own cell line, our experts can engineer the TARGATT™ system into your cell line. To learn more, explore our Custom TARGATT™ Master Cell Line Generation page or contact us today to schedule your free consultation.
Explore ASC’s series of antibody services and discover how we can help you propel your research forward. Whether you are looking to generate monoclonal antibodies or construct a cell-based library for screening, we have several well-established protocols and fully customizable service options for you to select from. Our single B cell antibody discovery platform is great for those seeking high-affinity rabbit monoclonal antibodies while our TARGATT™ HEK293 and CHO cell lines are ideal tools for researchers aiming to establish stable knock-in cell lines or large cell libraries. Visit our service pages or contact us today to learn more.
Advantages of the TARGATT™ Master Cell Lines
Advantages of ASC's Single B Cell Antibody Discovery Platform