Newsletter
iPSC-Dopaminergic Neurons
For several years, neuroscience research using cell line and animal models of degenerative neurological disorders such as Parkinson’s disease (PD) and Alzheimer’s has been plagued with the lack of translation of results to the human scenario, whether for basic understanding of disease pathology or for screening of targeted drug therapies. The development of the induced pluripotent stem cell (iPSC) technology and its further differentiation into various post-mitotic somatic cell lineages, especially neuronal lineage cells and dopaminergic neurons, has overcome a major bottleneck by providing biologically relevant in vitro models to screen drugs for neuroprotection and movement-disorder therapy.
ASC’s Human iPSC-derived Dopaminergic Neurons are derived from footprint-free, karyotype normal human iPSCs, ASE-9211 using a straightforward, optimized differentiation protocol, TH positive. We provide also provide high-purity mature dopamine neurons from our female iPSC line.
Advantages of using ASC’s iPSC-derived Dopaminergic Neurons:
- Dopamine neurons that are positive for dopamine marker
- Differentiated using footprint-free, integration-free protocols
- An ideal model for disease modeling, co-culture assays, neuroprotection, and neurotoxicity screening assays
- GMP iPSC Products & Services >> Learn More
If you have an iPSC line and would like to differentiate your cells into dopaminergic neurons, we offer custom iPSC dopaminergic differentiation services. Our team of scientists can even help you develop the downstream assay you need to move your research forward.
Products and Services
Case Studies
Case Study 1
Characterization of iPSC-derived Dopaminergic Neurons (ASE-9742)
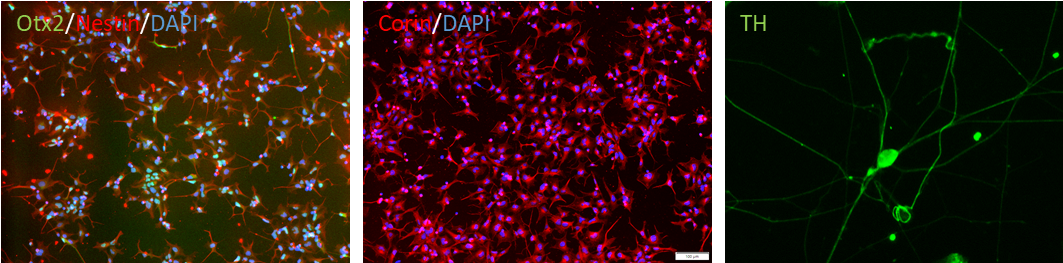
Figure 1. Immunostaining of the ASE-9742 Dopaminergic Neurons derived from the human iPSC control
line ASE-9211. The dopaminergic neuron precursors (provided separately) were identified by Otx2, Nestin and
Corin (Left, Middle). The mature dopaminergic neurons were identified by TH (Right).
Application Notes
- Controls for predictive models of neurological disorders
- Drug screening
- Neuroprotection assays and research
- Neurotoxicity screening
Publications
Shaltouki, A., Sivapatham, R., Pei, Y., Gerencser, A. A., Momčilović, O., Rao, M. S., & Zeng, X. (2015). Mitochondrial alterations by PARKIN in dopaminergic neurons using PARK2 patient-specific and PARK2 knockout isogenic iPSC lines. Stem cell reports, 4(5), 847-859.
Efthymiou, A. G., Steiner, J., Pavan, W. J., Wincovitch, S., Larson, D. M., Porter, F. D., ... & Malik, N. (2015). Rescue of an in vitro neuron phenotype identified in Niemann-Pick disease, type C1 induced pluripotent stem cell-derived neurons by modulating the WNT pathway and calcium signaling. Stem cells translational medicine, 4(3), 230-238.
Efthymiou, A., Shaltouki, A., Steiner, J. P., Jha, B., Heman-Ackah, S. M., Swistowski, A., ... & Malik, N. (2014). Functional screening assays with neurons generated from pluripotent stem cell–derived neural stem cells. Journal of biomolecular screening, 19(1), 32-43.
Shaltouki, A., Peng, J., Liu, Q., Rao, M. S., & Zeng, X. (2013). Efficient generation of astrocytes from human pluripotent stem cells in defined conditions. Stem cells, 31(5), 941-952.
FAQs
Can the iPSC-Dopaminergic Neurons be passaged?