Validation Data for Humanized PD-L1 Mouse
Strain Name: C57BL/6-Cd274em1(hPD-L1)/Asc Strain Background: C57BL/6
Programmed cell death ligand-1 (PD-L1) is a critical immune checkpoint molecule targeted for immunotherapies. This protein, also known as the cluster of differentiation 274 (CD274) or B7 homolog 1 (B7H1), is a transmembrane protein encoded by the CD274 gene and is the ligand for the PD-1 cell surface receptor. The binding of PD-L1 to PD-1 expressed on activated T cells transmits an inhibitory signal that inactivates cytotoxic T cells. This inhibitory mechanism is leveraged by tumor cells which overexpress PD-L1 and thereby inhibit the function of tumor-infiltrating T cells in order to escape immune surveillance. The humanized PD-L1 mouse model enables better translational results to test the efficacy of anti-PD-L1 immunotherapies.
Humanized Strategy
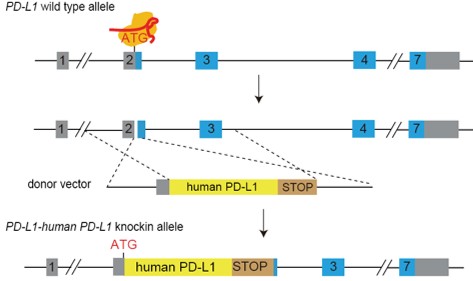
Figure 1. Generation strategy of humanized PD-L1 mice. The full-length protein coding sequence for human PD-L1 was placed immediately downstream of the start codon of the mouse endogenous Pd-l1 gene, followed by a poly(A) site, so that the expression of endogenous Pd-l1 in the mouse was replaced by the expression of fully humanized PD-L1 protein. This mouse model was generated in the C57BL/6J genetic background.
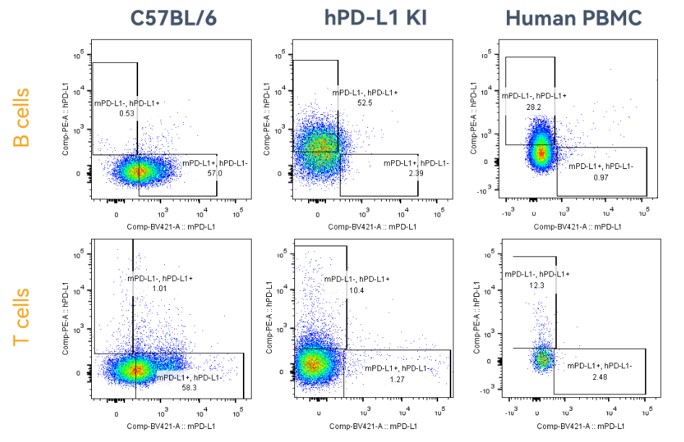
Figure 2. Expression of PD-L1 in the spleen lymphocytes collected from homozygous humanized PD-L1 mice and wild-type mice is detected by FACS. The results showed that the expression of human PD-L1 can be detected in both T cells and B cells collected from the spleen of homozygous humanized PD-L1 mice. (Data in partnership with collaborators)
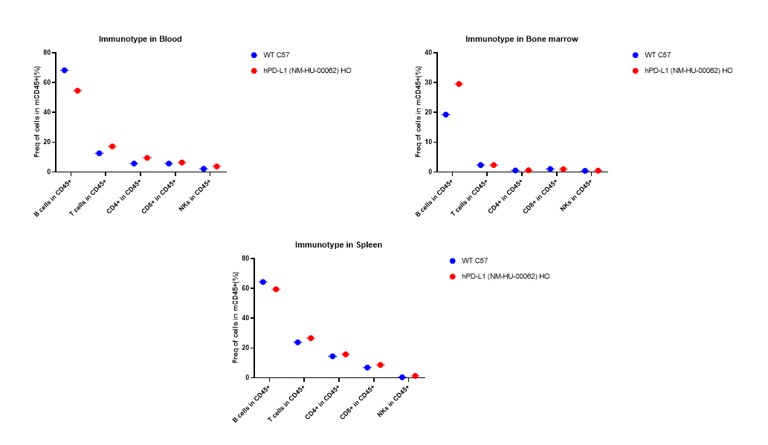
Figure 3. Immunotype in blood , spleen and bone marrow in hPD-L1 mice.
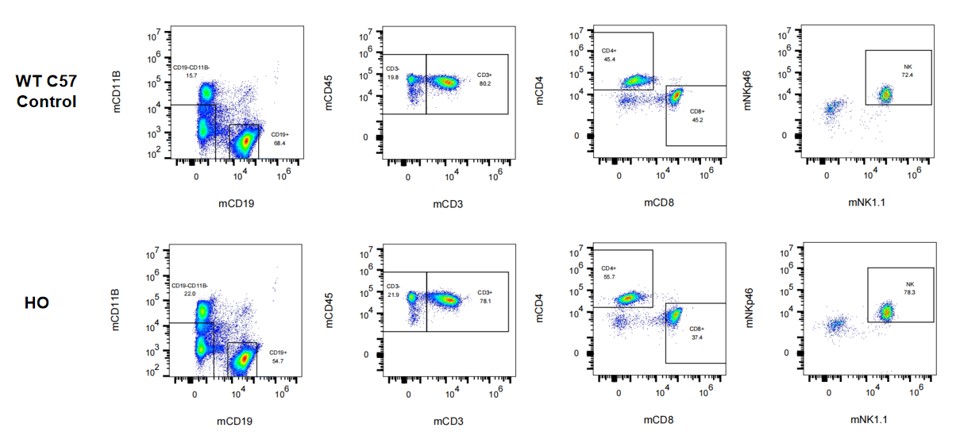
Figure 4. Immunotype in blood in hPD-L1 mice.
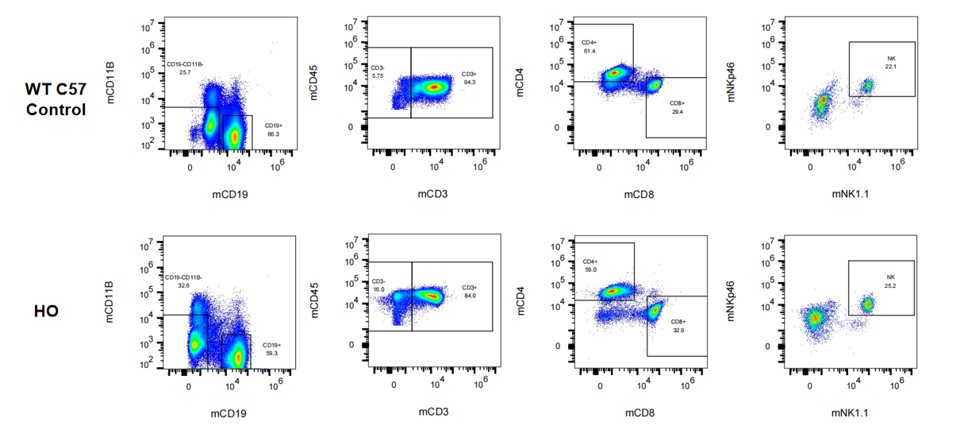
Figure 5. Immunotype in spleen in hPD-L1 mice.
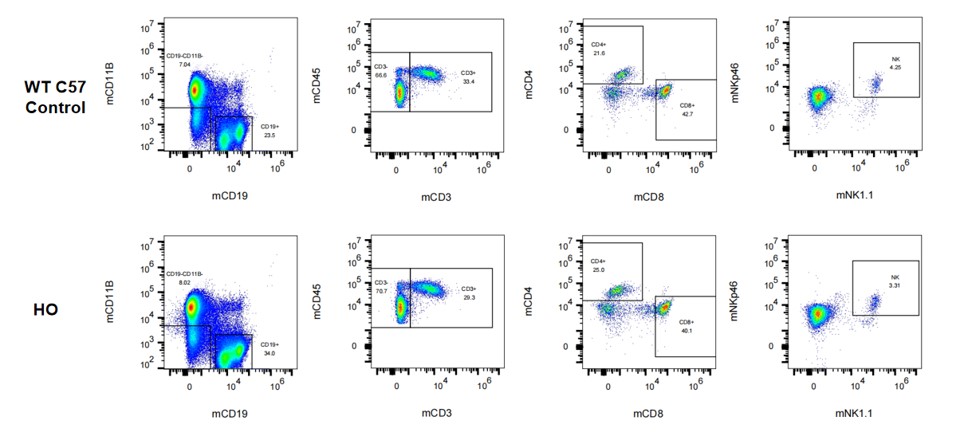
Figure 6. Immunotype in bone marrow in hPD-L1 mice.
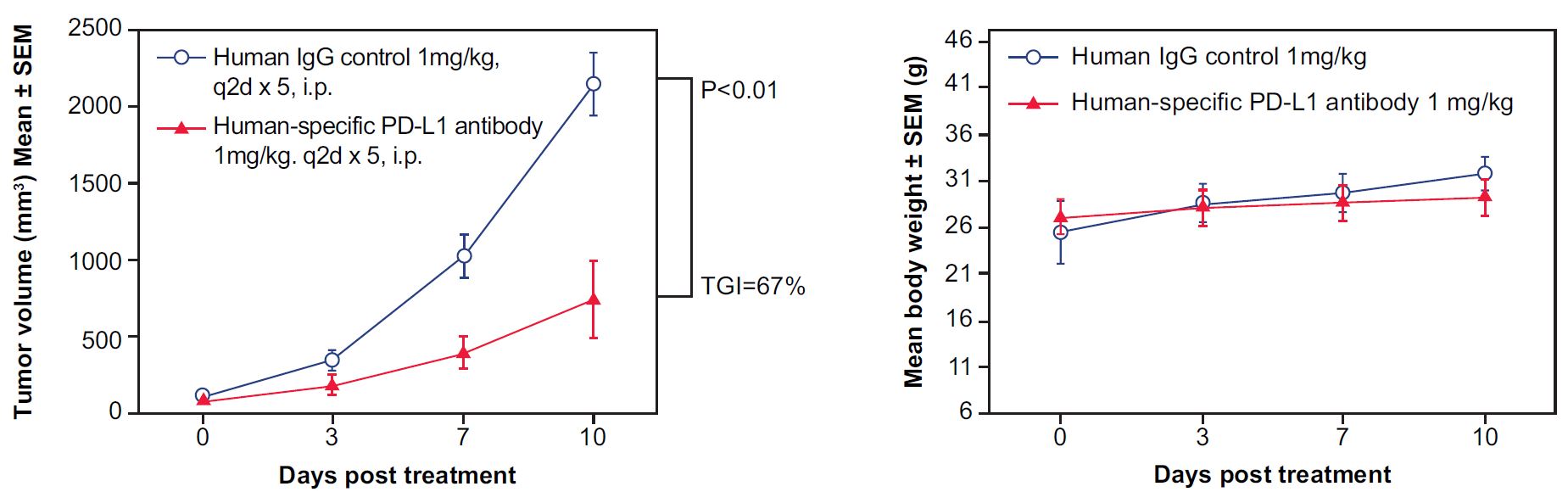
Figure 7. In vivo validation of anti-tumor efficacy in a MC38 tumor-bearing model of humanized PD-L1 mice. Mouse colon cancer cells MC38 engineered to express human PD-L1 were implanted subcutaneously into homozygous, humanized PD-L1 mice. The mice were randomly assigned into two groups when the tumor volume reached 100 mm3, with one group receiving human IgG as a control and the other receiving a human-specific, PD-L1 antibody (n=5). The human PD-L1 blocking antibody significantly inhibited tumor growth in the homozygous humanized mice (Left) without affecting overall body weight (Right), suggesting that the humanized PD-L1 mice represent an ideal model for evaluating the efficacy of therapeutic antibodies targeting human PD-L1. (TGI: tumor growth inhibition; 67%; p < 0.001), demonstrating that the humanized PD-L1 mice are a good in vivo model for validating the efficacy of antibodies targeting human PD-L1.