Humanized Strategy
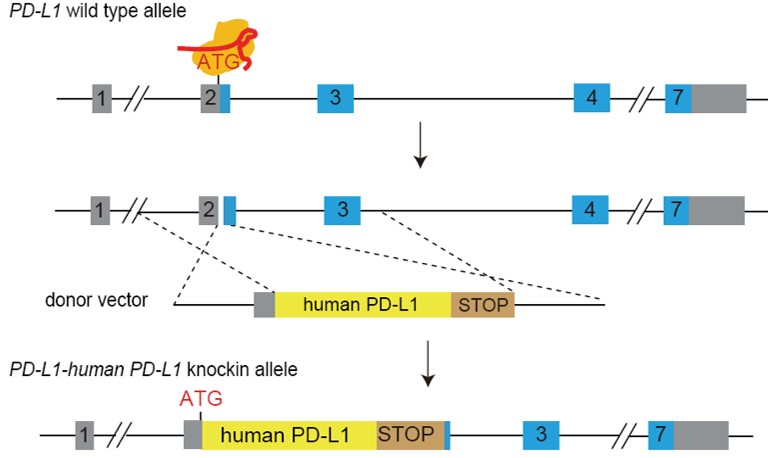
PD-L1 Humanization Strategy
4-1BB (2) Humanization Strategy
Validation Data: PD-L1
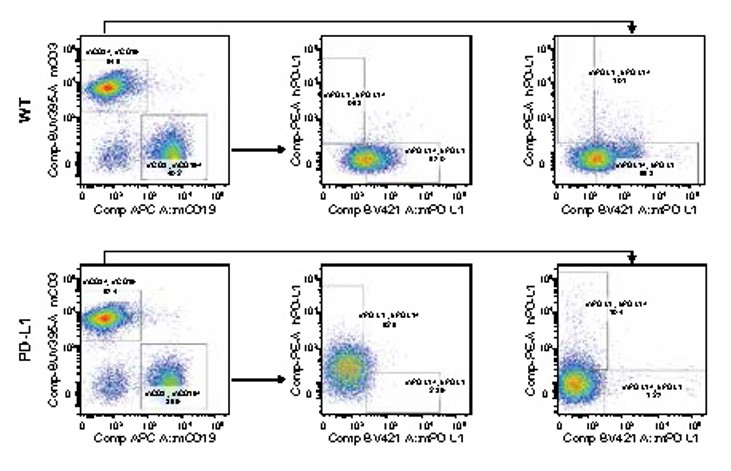
Figure 1. Expression of PD-L1 in the spleen lymphocytes collected from homozygous humanized PD-L1 mice and wild-type mice is detected by FACS. The results showed that the expression of human PD-L1 can be detected in both T cells and B cells collected from the spleen of homozygous humanized PD-L1 mice. (Data in partnership with collaborators)
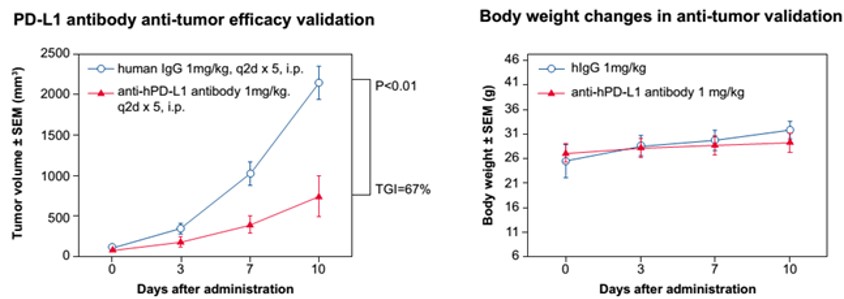
Figure 2. In vivo validation of anti-tumor efficacy in a MC38 tumor-bearing model of humanized PD-L1 mice. Homozygous humanized PD-L1 mice were inoculated with MC38 colon cancer cells (expressing human PDL1 rather than murine PD-L1). After the tumors grew to 100 mm3, the animals were randomly assigned into a control group and a treatment group (n=5). The results showed: The antibodies targeting human PD-L1 were associated with a very significant anti-tumor effect (TGI: tumor growth inhibition, p < 0.001), demonstrating that the humanized PD-L1 mice are a good in vivo model for validating the efficacy of antibodies targeting human PD-L1.
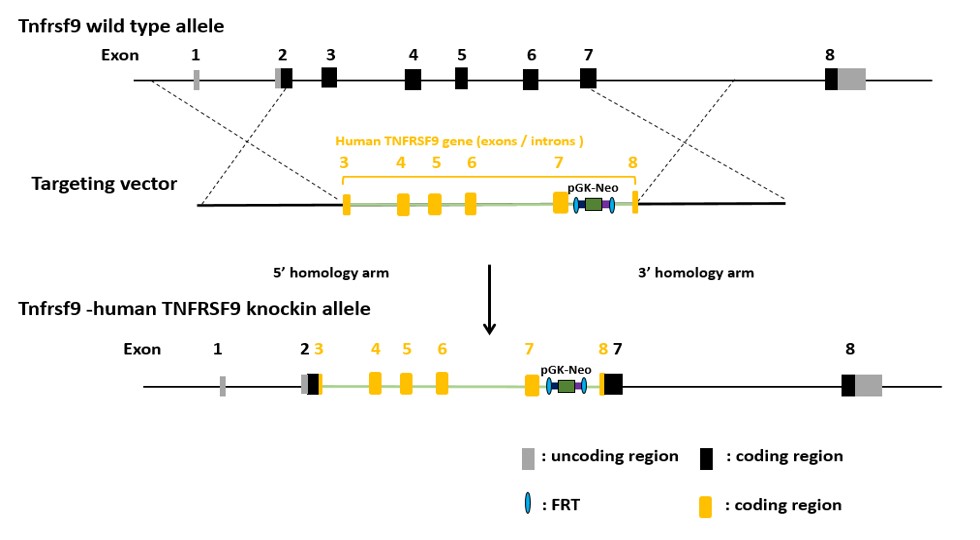
Validation Data: 4-1BB (2)
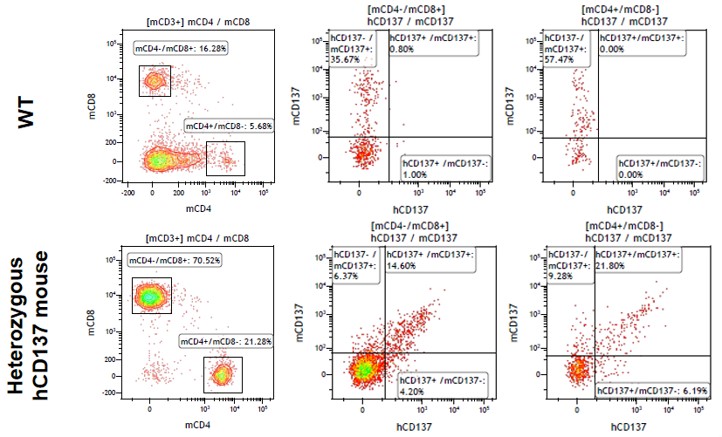
Figure 3. Expression of humanized CD137(4-1BB) in the activated spleen lymphocytes of heterozygous humanized 4-1BB mice is detected by FACS.
The spleen lymphocytes of heterozygous humanized 4-1BB mice were activated by anti-CD3 and anti-CD28 for 48 hours, and then collected for staining. The expression of humanized 4-1BB was detected by FACS. The results showed that the active expression of humanized 4-1BB can be detected in both activated CD4+ and CD8+ T lymphocytes collected from heterozygous humanized 4-1BB mice. (Data in partnership with collaborators)
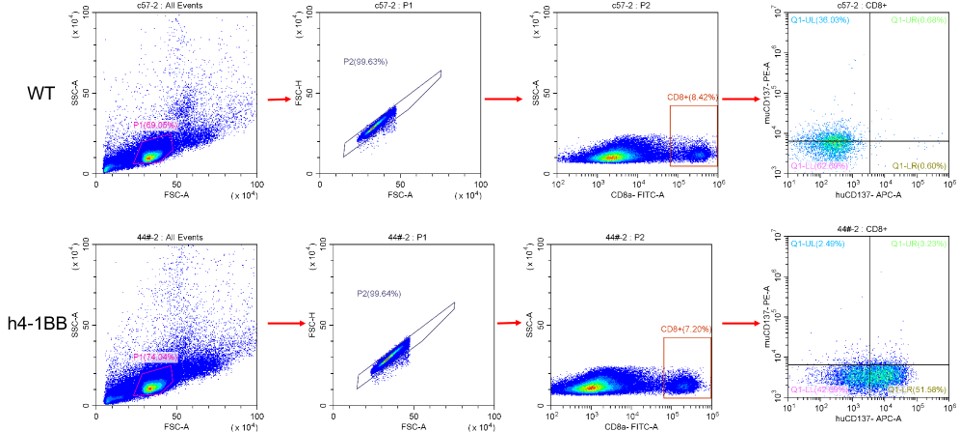
Figure 4. Analysis of human 4-1BB (CD137) expression in the spleen by FACS. The homozygous KI mouse expresses human 4-1BB on the CD8+ T cells.
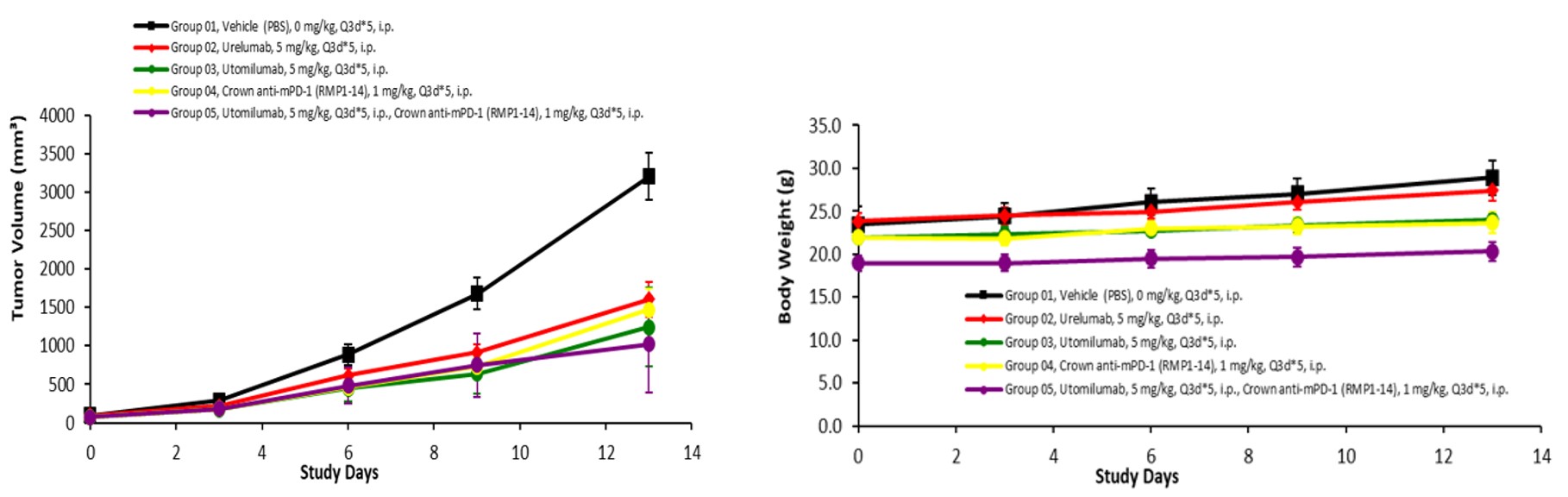
Figure 5. In vivo validation of humanized 4-1BB mice. Humanized 4-1BB mice were inoculated with MC38 colon cancer cells, and were randomly assigned into different groups (n=6) when the tumors grew to a volume of 100 mm3. Human-specific, 4-1BB antibody (Urelumab or Utomilumab) was given every three days (Q3D) for two weeks (Data in partnership with collaborators).


