PD-1 Knock-in Strategy
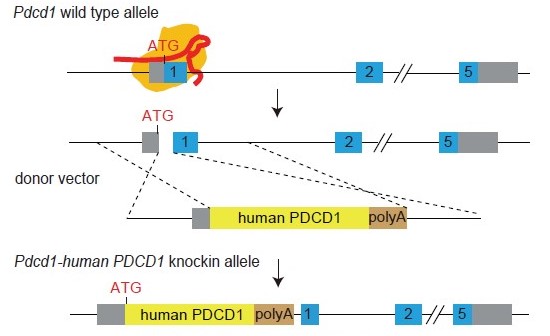
Figure 1. Gene knock-in strategy to engineer a humanized PD-1 (PD-1-HU) mouse model. The endogenous mouse Pdcd1 gene was replaced by insertion of the full length protein coding sequence for the human PDCD1 (PD-1) gene into the ATG position of mPdcd1, followed by a transcription terminator signal (polyA).
Human PD-1 mRNA and Protein Validation
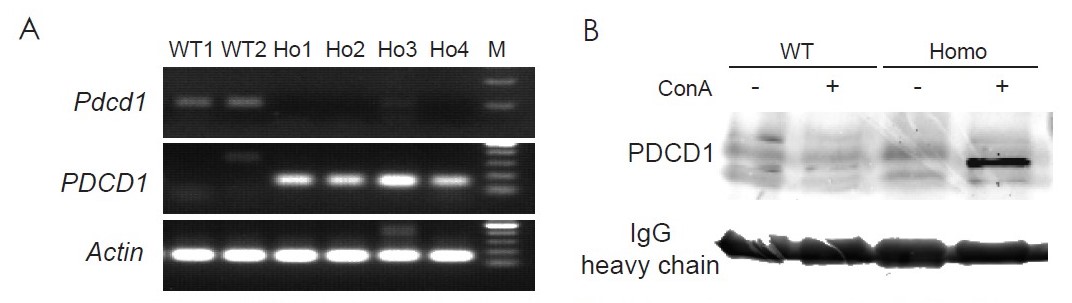
Figure 2. The humanized PD-1 mouse model (PD-1-HU) expresses human PDCD1 (PD-1) gene but not the mouse Pdcd1. Human PD-1 mRNA and protein expression was determined by qPCR (A) and Western Blot (B). A. the wildtype C57BL/6 mice (WT) expressed mouse Pdcd1 mRNA but not human PDCD1, while the humanized PD-1 (homozygous) mice showed expression of the human version of the PD-1 gene. (B) Human PD-1 expression was expressed only in homozygous, humanized PD-1 mice, after concavalin A (ConA) activation.
Human PD-1 Expression in Spleen T Cells After Stimulation
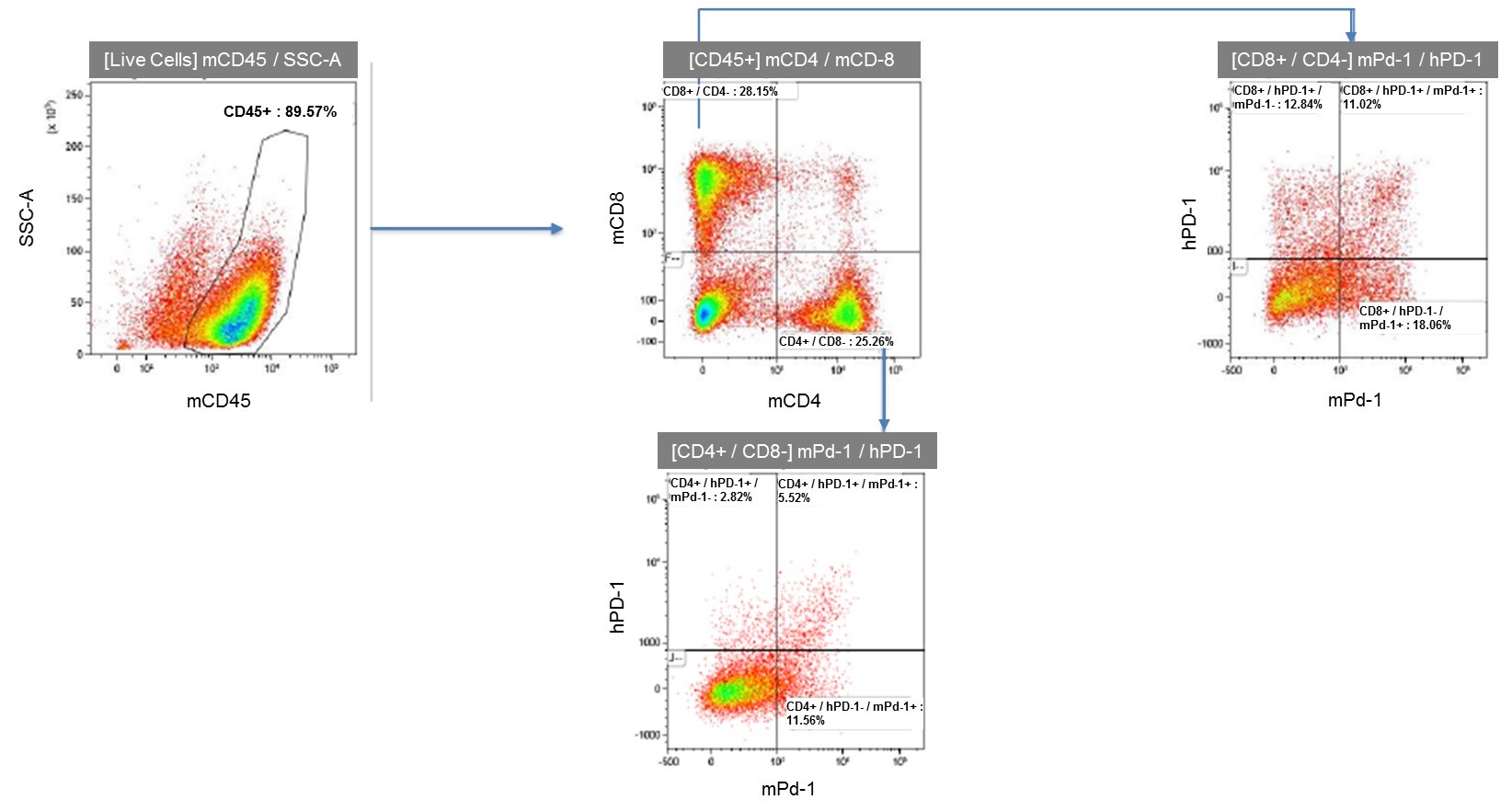
Figure 3. Flow cytometry analysis of activated spleen lymphocytes from humanized PD-1 homozygous mice shows expression of human PD-1.
In Vivo Validation of PD-1-HU Mouse Model in MC38 Colon Cancer Cell Line
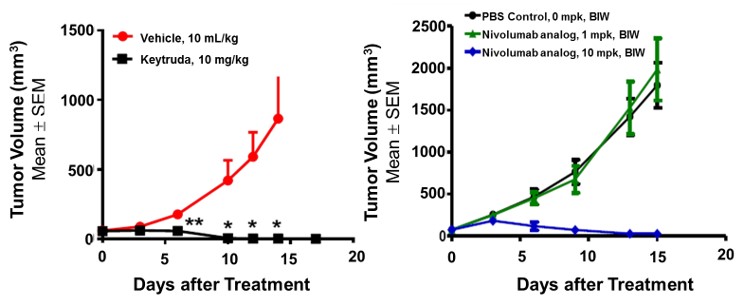
Figure 4. In vivo validation of the anti-tumor efficacy of immune checkpoint inhibitors, Keytruda and a Nivolumab analog in a MC38 tumor-bearing humanized PD-1 mouse model. Homozygous humanized PD-1 mice were inoculated with MC38 colon cancer cells. After the tumors grew to 100 mm3, the animals were randomly assigned to control and treatment groups (N=8). The drugs were given according to standard procedures established in the lab. The results showed a significant dose-dependent anti-tumor effect of the monoclonal antibodies targeting human PD-1 in this humanized mouse model demonstrating that the PD-1-HU humanized mouse model is an ideal in vivo model for validating efficacy of immunotherapies targeting human PD-1. Values are Mean ± SEM of tumor volume.
TIGIT Knock-in Strategy
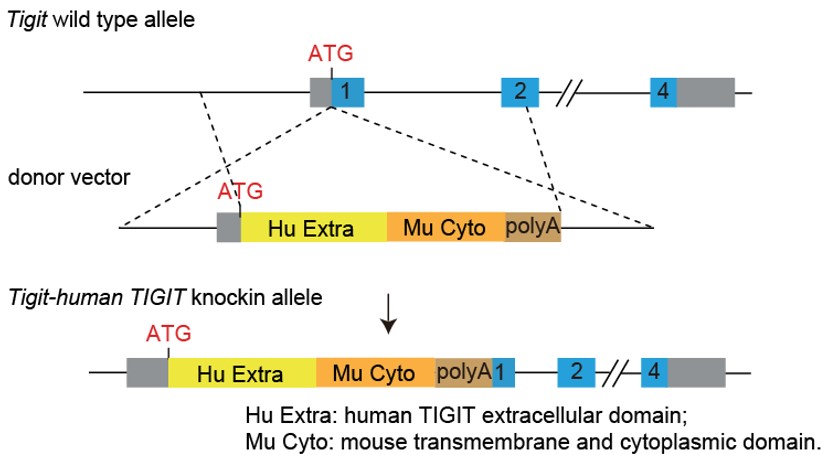
Figure 1. Generation strategy of humanized TIGIT mice. Under the C57BL/6J genetic background, the coding sequences for the extracellular domain of human TIGIT and the transmembrane and intracellular domains of murine TIGIT were inserted into the ATG position of the mouse TIGIT gene, so that the expression of endogenous TIGIT in the mouse was replaced by the expression of humanized chimeric protein.
TIGIT Expression on Polarized CD4+ T Cell
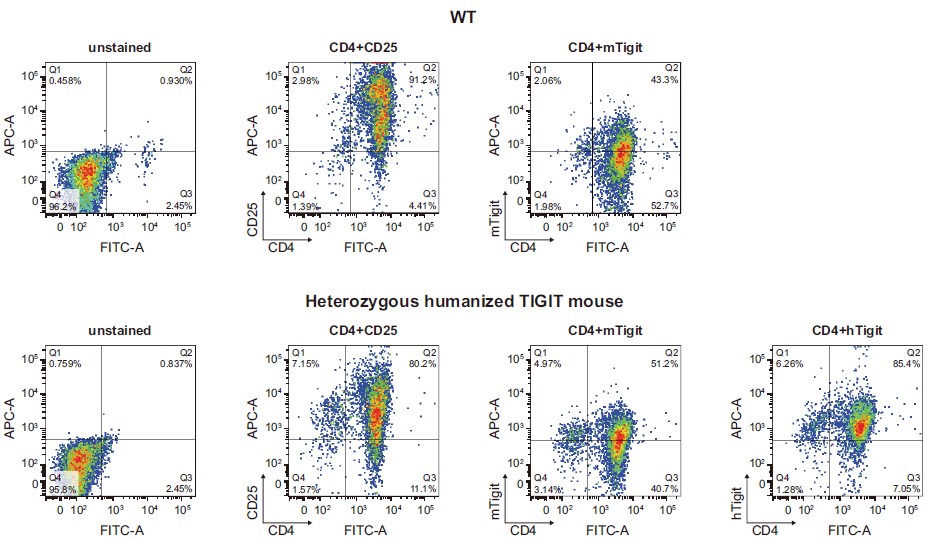
Figure 2. The expression of human TIGIT in the polarized CD4+ T cells derived from humanized TIGIT mice was confirmed by FACS. Naive spleen CD4+ T cells were isolated from heterozygous humanized TIGIT mice. After in vitro stimulation, activation and expansion by cytokines and antibodies, the CD4+ T cells were re-stimulated with PMA/ionomycin, followed by the measurement of human TIGIT expression in the polarized CD4+ T cells by FACS. The results demonstrated an active expression of human TIGIT in polarized CD4+ T cells derived from heterozygous humanized mice, with a comparable expression level to the endogenous murine Tigit.
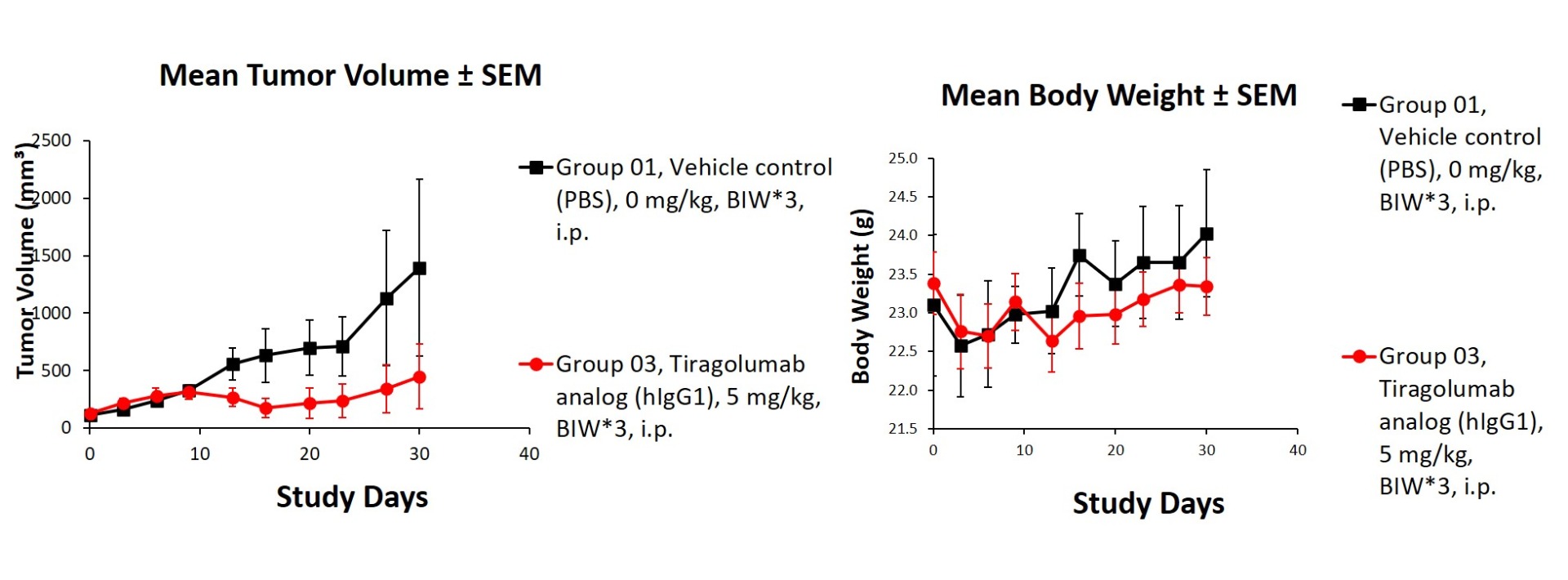
Figure 3. In vivo validation of anti-tumor efficacy in a Hepa1-6 tumor-bearing model of humanized TIGIT mice. Homozygous humanized TIGIT mice were inoculated with Hepa1-6 cells. After the tumors grew to 110 mm3, the animals were randomly assigned into a control group and a treatment group. The results showed a significant anti-tumor effect was observed when the antibody targeting human TIGIT. (Data in partnership with collaborators).
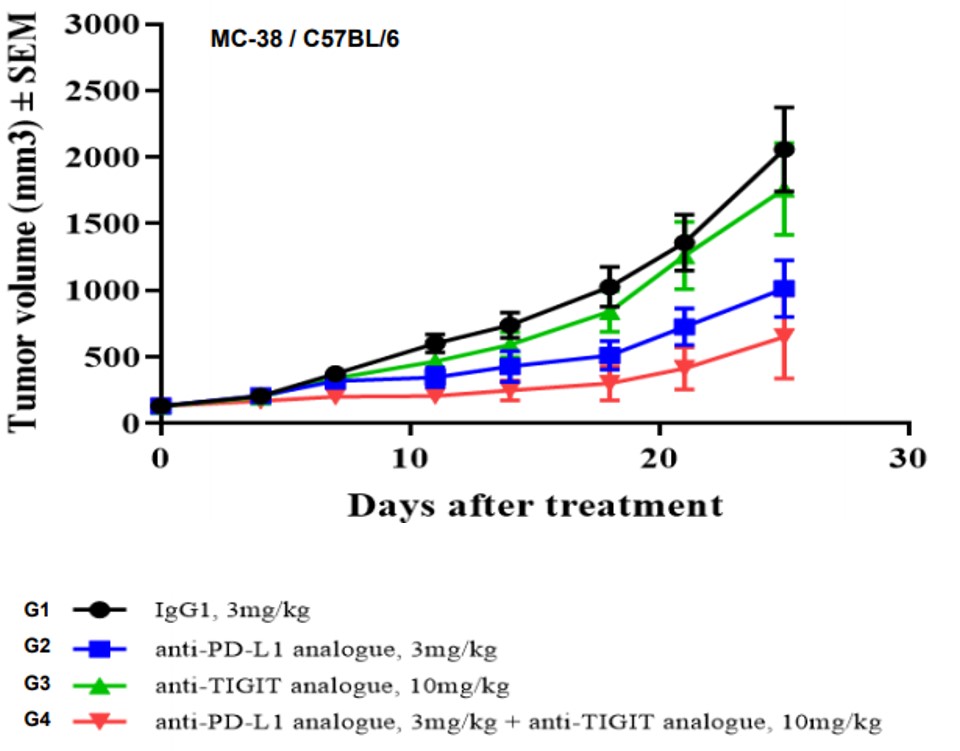
Figure 4. In vivo validation of anti-tumor efficacy in a MC38 tumor-bearing model of humanized TIGIT mice. Homozygous humanized TIGIT mice were inoculated with MC38 colon cancer cells. After the tumors grew to 130 mm3, the animals were randomly assigned into a control group and a treatment group (n=7). The results showed a significant anti-tumor effect was observed when the antibody targeting human TIGIT was administered together with Anti-PD-L1 analogue. (Data in partnership with collaborators).
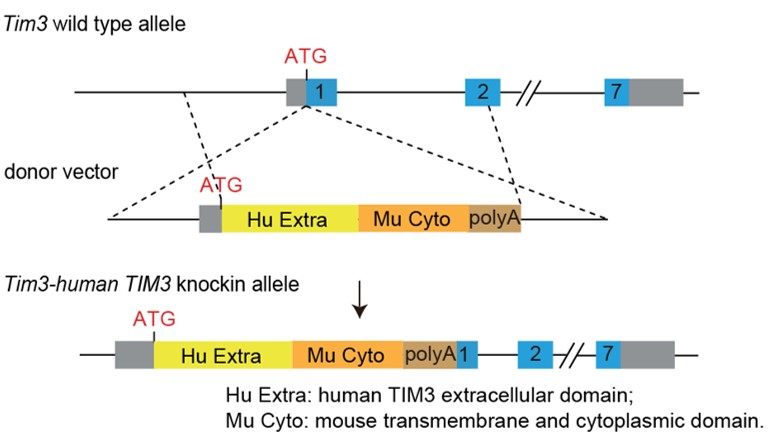
TIM3 Knock-in Strategy
FACS Analysis of Human TIM3 Expression on Stimulated Mouse T Cells
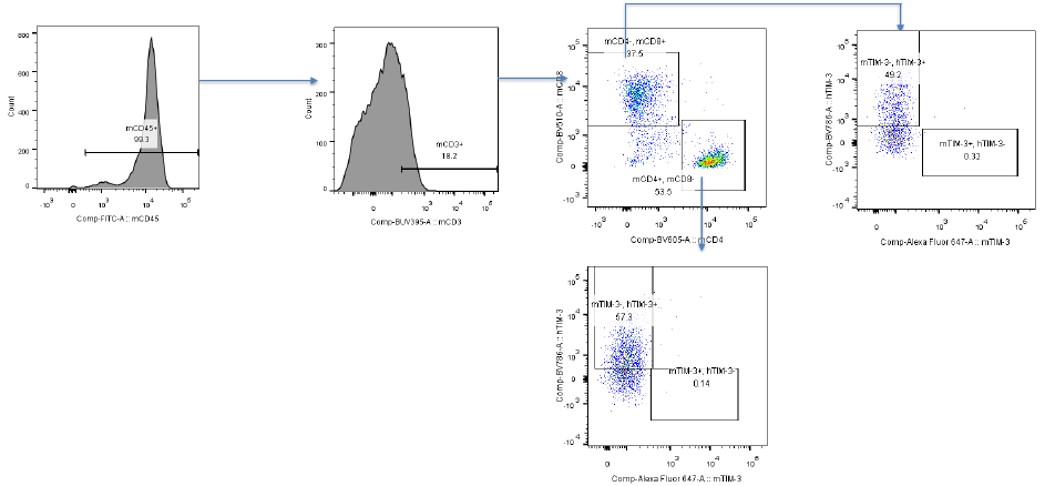
Data in partnership with collaborators


